Endotoxin Removal Technology
Fundamentals and Representative Removal Methods
April 02, 2026
In pharmaceuticals, medical devices, regenerative medicine, and research applications, the control and removal of endotoxins are critical issues that directly affect product safety and quality. However, endotoxins exhibit high resistance to heat and chemical agents and, in many cases, cannot be sufficiently addressed by conventional sterilization processes alone.
This article provides an overview of the fundamental characteristics of endotoxins, outlines representative endotoxin removal methods, and explains the key features and considerations of each approach.
What Are Endotoxins?
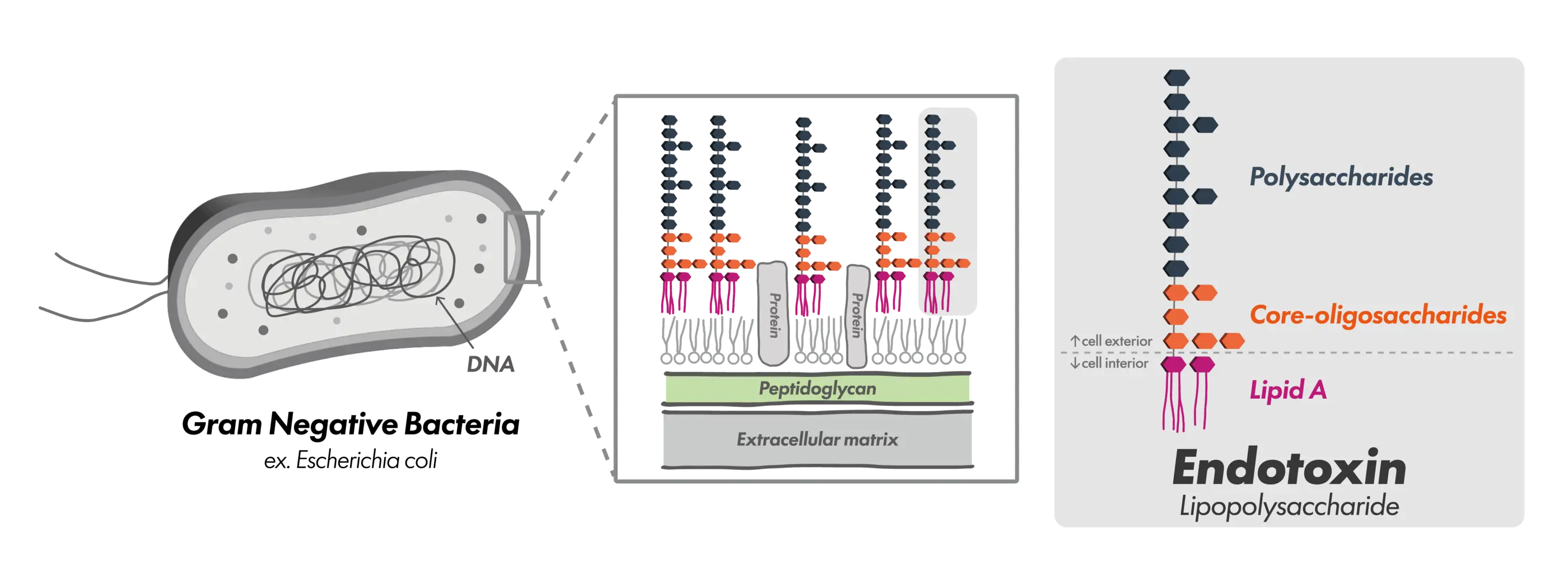
Endotoxins are lipopolysaccharides (LPS) that constitute the outer membrane of Gram-negative bacteria and are biologically active substances released during bacterial growth, as well as upon cell death or disruption. In particular, the lipid moiety known as Lipid A is the primary determinant of endotoxin biological activity.
Endotoxins are widely distributed in the environment and can be found in a variety of settings, including tap water, soil, and even the air.
It is well known that even trace amounts of endotoxins can induce strong febrile and inflammatory responses in humans. For this reason, strict control of endotoxins is required in applications such as injectable pharmaceuticals, medical devices, and regenerative medicine.
In addition, endotoxins exhibit high resistance to heat and chemical agents, making them difficult to inactivate by conventional sterilization processes. Consequently, unlike simple microbial removal, dedicated endotoxin reduction strategies are required.
Importance of Endotoxin Removal
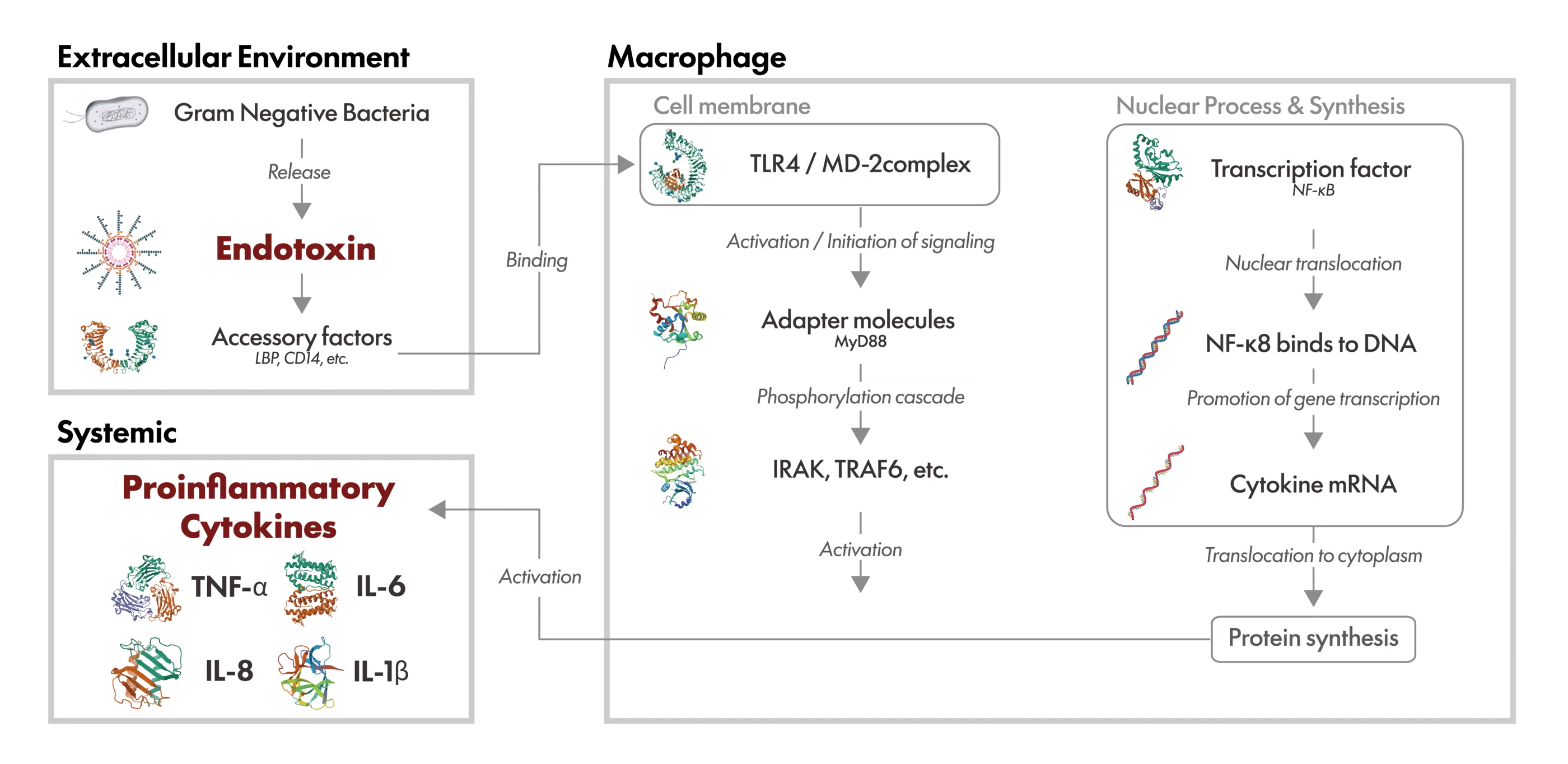
If endotoxins are not adequately removed and remain in pharmaceuticals, medical devices, cell culture–related products, cell therapy products, or regenerative medicine products, there is a risk of inducing serious adverse biological reactions in humans.
Reported symptoms include fever, chills, inflammatory responses, and hypotension, while severe cases may progress to multiple organ failure or septic shock. These effects occur because immune cells, such as macrophages, recognize endotoxins as foreign substances and transmit intracellular signals via Toll-like receptor 4 (TLR4), leading to excessive production of pro-inflammatory cytokines, including TNF-α, IL-1β, IL-6, and IL-8.
In particular, for injectable drugs and medical devices that come into direct contact with blood, even extremely small amounts of endotoxins may lead to serious adverse health effects.
Because endotoxins are difficult to completely inactivate by standard sterilization processes, there are cases in which products meet sterility test requirements yet still contain residual endotoxins, resulting in insufficient safety assurance. Therefore, from the perspectives of product quality and patient safety, the design and control of manufacturing processes that reliably remove endotoxins are essential.
Fields Requiring Endotoxin Control
Endotoxin control is particularly important in fields that handle products that directly or indirectly affect the human body.
| Field | Typical Products | Risks and Challenges | Key Control Considerations |
|---|---|---|---|
| Pharmaceuticals | Injectable drugs Infusion solutions Dialysis fluids Vaccines | Direct entry into the bloodstream means that even trace levels of endotoxins can cause serious adverse reactions, including fever, shock, and multiple organ failure. | Strict assurance of sterility and pyrogen (endotoxin)-free conditions throughout the entire manufacturing process, from process water to the final product. |
| Medical Devices | Catheters Implants Artificial organs Hemodialysis circuits | Direct contact with blood or cerebrospinal fluid creates a risk that surface contaminants may leach into the body. | In addition to ensuring product cleanliness, validation of cleaning processes and prevention of recontamination during sterile packaging are essential. |
| Regenerative Medicine/Cell Therapy | Cell-processed products Culture media Culture vessels | Because the products consist of living cells, terminal heat sterilization is not feasible. Contamination can result in product disposal or adverse health effects. | As terminal sterilization is not possible, thorough endotoxin removal at the raw material stage (e.g., media and reagents) is the only effective approach. |
| Research and Testing | Immune cell assays Differentiation studies | Endotoxin contamination can cause unintended cell activation or cell death, leading to unreliable experimental results. | To ensure the validity and reproducibility of experimental data, endotoxin-free reagents and materials are required. |
In these fields, simply removing microorganisms is not sufficient; reliable reduction and control of endotoxins are critical factors that determine product safety and quality.
Endotoxin Control Standards
In fields where endotoxin control is required, endotoxin limits and control standards are defined by pharmacopeias such as the Japanese Pharmacopoeia (JP) and the United States Pharmacopeia (USP).
| Category | Application | Endotoxin Limit | Primary Reference |
|---|---|---|---|
| Pharmaceuticals | General injectable products | 5.0 EU/kg | JP 4.01 / USP <85> |
| Intrathecal administration | 0.2 EU/kg | JP 4.01 / USP <85> | |
| Medical Devices | General (blood-contacting) | < 20 EU/device | USP <161> |
| Cerebrospinal fluid–contacting | < 2.15 EU/device | USP <161> | |
| Regenerative Medicine/Cell Therapy | Water for injection | < 0.25 EU/mL | JP monographs / USP <1231> |
| Dialysis water | < 0.050 EU/mL | ISO / JSDT | |
| Research and Testing | Cell culture (reference range) | < 0.01∼0.1 EU/mL | Internal or supplier-specific standards |
To meet these endotoxin control standards, two main approaches are used: methods that degrade or inactivate endotoxins and methods that physically separate endotoxins from the target material.
Methods for Removing Endotoxins by Degradation
Methods for removing endotoxins by degradation (destruction) involve physically or chemically altering the structure of lipopolysaccharides (LPS) to eliminate their biological activity. This section describes representative approaches, such as heat treatment, chemical treatment, and radiation exposure, with a focus on the mechanisms of endotoxin inactivation. (Note: This discussion includes general methods that are not listed in JP or the USP.)
Dry Heat Treatment
Endotoxins possess a lipopolysaccharide (LPS) structure composed of Lipid A, a core oligosaccharide, and an O-antigen. Their biological activity, particularly pyrogenic activity, is primarily attributed to the Lipid A moiety.
Dry heat treatment involves exposure to high temperatures, typically 250 °C for 30 minutes or longer, and relies on oxidative degradation induced by thermal energy. Under these conditions, ester linkages and intramolecular structures within Lipid A are disrupted, leading to irreversible changes in its three-dimensional structure. As a result, endotoxins lose their ability to bind to human immune receptors, specifically the TLR4/MD-2 complex, and their activity is significantly reduced.
※ It should be noted that conventional autoclave sterilization using moist heat (e.g., 121 °C) does not readily induce this type of degradation reaction, and therefore endotoxins are generally not inactivated under standard autoclaving conditions.
Strong Alkaline Treatment
Strong alkaline treatment is a method that utilizes chemical saponification reactions under highly alkaline conditions, typically using strong bases such as sodium hydroxide (NaOH).
Under alkaline conditions, ester bonds within Lipid A undergo hydrolysis, resulting in cleavage of the fatty acid side chains. This alters the three-dimensional structure of Lipid A, leading to loss of its ability to bind to human immune receptors, specifically the TLR4/MD-2 complex, thereby inactivating the endotoxin.
In addition, alkaline conditions can fragment the polysaccharide portion of the molecule, increasing its overall water solubility. This facilitates the physical removal of endotoxin during subsequent washing or rinsing steps, an additional advantage of this approach.
| NaOH Concentration | Treatment Temperature | Treatment Time |
|---|---|---|
| 1.0 M | Room temperature | 1–2 hours |
| 0.5 M | Room temperature | 4–12 hours |
| 0.1 M | Room temperature | ≥16 hours |
| 0.5 M + EtOH* | Room temperature | 30–60 minutes |
*The use of organic solvents such as ethanol can enhance penetration into highly hydrophobic LPS, which may allow for a reduction in treatment time.
Oxidative Treatment
Oxidative treatment employs strong oxidizing agents such as hydrogen peroxide, hypochlorous acid, and ozone, particularly those that generate highly reactive hydroxyl radicals (•OH). These agents oxidize the fatty acid chains and polysaccharide components of Lipid A, leading to cleavage of intramolecular bonds.
As a result, the three-dimensional structure of LPS is disrupted, preventing interaction with human immune receptors such as the TLR4/MD-2 complex and consequently eliminating its biological activity.
Low-Temperature Hydrogen Peroxide Gas Plasma Treatment
Low-temperature hydrogen peroxide gas plasma treatment is a method in which hydrogen peroxide vapor is converted into a plasma, and the resulting highly reactive species (radicals) and ultraviolet (UV) radiation are used for treatment. In addition to chemical oxidation by reactive species, physical “etching” effects caused by ions within the plasma also contribute to the process.
Through these combined effects, the fatty acid chains of Lipid A and the polysaccharide structures are degraded and removed. In hydrogen peroxide/peracetic acid plasma systems, the strong oxidative power of peracetic acid is additionally applied, which is expected to enable more efficient endotoxin inactivation.
Electron Beam and Gamma Irradiation
Electron beam (EB) and gamma irradiation are widely used sterilization methods that inactivate microorganisms by damaging their DNA and RNA through high-energy radiation. However, their effectiveness against endotoxins varies significantly depending on treatment conditions, particularly the presence or absence of moisture.
Because Lipid A, the active center of endotoxins, exhibits extremely high resistance to radiation compared with DNA and RNA, conventional sterilization doses (approximately 25 kGy) are insufficient to adequately inactivate endotoxins in the dry state. In contrast, in aqueous solutions, radicals generated by the radiolysis of water (indirect effects) can attack LPS, allowing degradation to proceed even at relatively lower irradiation doses.
| Purpose | State | Typical Irradiation Dose | Effect |
|---|---|---|---|
| Sterilization (Sterility assurance) | Typical | 15–25 kGy | Destruction of DNA/RNA achieves the sterility assurance level (SAL10-6). However, endotoxins are likely to remain. |
| Endotoxin degradation | Dry | >100 kGy | Because only direct radiation effects are involved, unrealistically high doses are required to disrupt endotoxin structure, at levels that can cause material degradation. |
| Aqueous solution | 10–50 kGy | Hydroxyl radicals (•OH) generated by water radiolysis oxidatively degrade Lipid A via indirect radiation effects. |
High-Pressure Steam (Autoclave) Treatment
High-pressure steam treatment is a moist heat process that uses high-temperature saturated steam and is widely employed for microbial sterilization, typically at 121 °C for 15–20 minutes. However, to inactivate endotoxins using moist heat, much more severe conditions are required, such as exposure at 121 °C for 5 hours or longer, or at 130–140 °C for several hours, far exceeding standard sterilization conditions.
In the presence of moisture and sufficient thermal energy, bonds within the polysaccharide regions and Lipid A undergo hydrolysis, resulting in changes to the molecular structure. Consequently, the three-dimensional structure of LPS can no longer be maintained, and the structural features required for binding to human immune receptors, such as the TLR4/MD-2 complex, are lost.
However, these conditions are excessively harsh not only for pharmaceutical products but also for materials such as rubber and plastics, making them prone to hydrolysis and thermal denaturation. As a result, this approach is generally impractical as a method for endotoxin degradation.
Enzymatic Degradation
Enzymatic degradation is a method that inactivates endotoxins by using specific enzymes to decompose their constituent components. Unlike chemical reagents or high-temperature treatments, enzymatic degradation operates under relatively mild conditions, allowing endotoxins to be treated while minimizing damage to the target material.
However, enzymatic reactions are highly sensitive to factors such as solution pH, temperature, and coexisting substances, making it challenging to precisely control the reaction conditions required for complete endotoxin inactivation.
| Enzyme | Target Site | Reaction | Resulting Structural Change |
|---|---|---|---|
| Acyloxyacyl Hydrolase (AOAH) | Secondary acyl chains (fatty acids) | Deacylation (hydrolysis) | Hexa-acylated → Tetra-acylated* *A host defense mechanism present in human neutrophils and other immune cells; endotoxin activity is significantly reduced. |
| Alkaline Phosphatase (ALP) | Phosphate groups | Dephosphorylation (hydrolysis) | Diphosphoryl → Monophosphoryl* *Removal of a phosphate group weakens binding affinity to immune receptors. |
Methods for Removing Endotoxins by Separation
In endotoxin removal, in addition to methods that chemically or physically destroy (inactivate) endotoxin structures, approaches that physically separate and remove endotoxins while preserving their structural and physicochemical properties are also widely used.
Distillation
Distillation is a classical separation method in which a liquid is boiled and vaporized, followed by condensation to separate impurities. Because endotoxins have high molecular weights and are nonvolatile, they do not transfer to the vapor phase during the phase transition of water to steam but instead remain in the distillation residue. As a result, the condensed distillate contains little to no endotoxin.
This method is primarily used in the production of pharmaceutical-grade water, such as Water for Injection (WFI) and purified water, and in manufacturing processes involving volatile compounds.
Ultrafiltration and Reverse Osmosis
Ultrafiltration and reverse osmosis are separation methods that utilize membrane pore size and molecular sieving effects. In aqueous solutions, endotoxins exist not only as monomers but also form large aggregates (micelles) through hydrophobic interactions. As a result, endotoxins cannot pass through membranes with molecular weight cutoffs above a certain threshold. Consequently, only water molecules and low-molecular-weight components permeate the membrane, while endotoxins are retained on the upstream side.
| Endotoxin Monomer | Endotoxin Aggregate | |
|---|---|---|
| Molecular weight (Da) | 10,000–20,000 | 300,000–1,000,000 |
| Structure | Lipid A + core oligosaccharide + O-antigen (single-molecule state) | Hydrophobic regions associate in aqueous solutions to form large micelles or vesicle-like structures |

| Comparison Item | Ultrafiltration (UF) | Reverse Osmosis (RO) |
|---|---|---|
| Separation principle | Size exclusion (molecular sieving effect) | Diffusion and dissolution/rejection (dense membrane permeation driven by osmotic pressure difference) |
| Removal mechanism | Physical retention based on molecular size; removal of endotoxin micelles and monomers | Complete rejection based on molecular size and charge; removal of ions, organic compounds, and other solutes |
| Applicable processes | Purification of protein formulations and biopharmaceuticals | Production of pharmaceutical-grade water |
| Challenges/risks | In the presence of chelating agents or surfactants, endotoxin micelles may dissociate, creating a risk of leakage through membranes with larger molecular weight cutoffs. | Not suitable for solutions containing high-molecular-weight pharmaceuticals such as proteins, as the product itself would be removed. |
Surfactants
Endotoxins can be separated using surfactants (detergents), taking advantage of their amphiphilic nature. This approach can be broadly classified into two types: methods based on physical phase separation and methods based on chemical dissociation.
Cloud Point Extraction
Cloud point extraction is a method that exploits the property of certain nonionic surfactants, such as Triton X-114, to separate into an aqueous phase and a surfactant-rich phase in response to temperature changes, a phenomenon known as the cloud point. When the temperature is raised above the cloud point (approximately 22 °C), highly hydrophobic endotoxins are partitioned into the surfactant-rich phase, while proteins remain in the aqueous phase, enabling their separation.
Micelle Dissociation and Washing Method
This approach uses surfactants to forcibly dissociate the large aggregates (micelles) formed by endotoxins into monomeric species. By doing so, hydrophobic interactions between proteins and endotoxins are disrupted, making this method useful as a pretreatment step to enhance the efficiency of subsequent adsorption or membrane separation processes.
| Comparison Item | Cloud Point Extraction | Micelle Dissociation/Washing |
|---|---|---|
| Representative reagents | Nonionic surfactants Triton X-114 | Anionic surfactants: Sodium dodecyl sulfate (SDS), deoxycholate Nonionic surfactants: Polysorbate 20/80 |
| Separation principle | Phase separation Temperature increase (>22 °C) induces separation into two phases, partitioning highly hydrophobic endotoxins into the surfactant-rich phase | Monomerization Large endotoxin micelles are dissociated into monomers (10–20 kDa), disrupting binding to proteins |
| Advantages | Simple operation High protein recovery can be maintained | Enables physical disruption of hydrophobic interactions between proteins and endotoxins Effective for equipment cleaning |
| Disadvantages | ・Residual risk: Complete removal of Triton X-114 is difficult ・UV interference: Triton absorbs at 280 nm, complicating protein quantification ・Limited adoption under GMP conditions | ・Membrane permeation risk: Monomerized endotoxins may pass through UF membranes (e.g., 100 kDa cutoff) ・LER phenomenon: Residual surfactants can cause false-negative results in LAL assays, representing a major risk factor |
| Typical applications | Early-stage purification and research use (e.g., endotoxin removal from crude extracts) | Intermediate purification and washing steps (e.g., pretreatment prior to affinity adsorption) |
Adsorption
Adsorption-based methods separate endotoxins from target materials by exploiting interactions between the negative charge and hydrophobic regions of endotoxin molecules and ligands (functional groups) on the surface of adsorbent materials.
Unlike heat or degradation-based treatments, adsorption does not destroy the endotoxin structure itself. As a result, this approach is well-suited for protein formulations and biopharmaceuticals that are sensitive to heat or chemical agents and must be processed while preserving their original properties.
| Category | Adsorption Principle |
|---|---|
| Affinity columns | Biological affinity Utilize specific binding interactions between Lipid A and ligands such as polymyxin B |
| Anion exchange resins | Electrostatic interactions Negatively charged endotoxins are adsorbed onto positively charged resins |
| Depth filters | Zeta potential (positive charge) Membrane surfaces are cationically modified to adsorb and retain endotoxins via electrostatic attraction |
| Synthetic adsorbents | Specific interactions Utilize affinity for particular amino acid sequences or functional groups |
Another key feature of adsorption-based methods is the flexibility to select different operational formats, such as column-based or batch-based processes, depending on processing scale and application requirements.
Column-Based Method
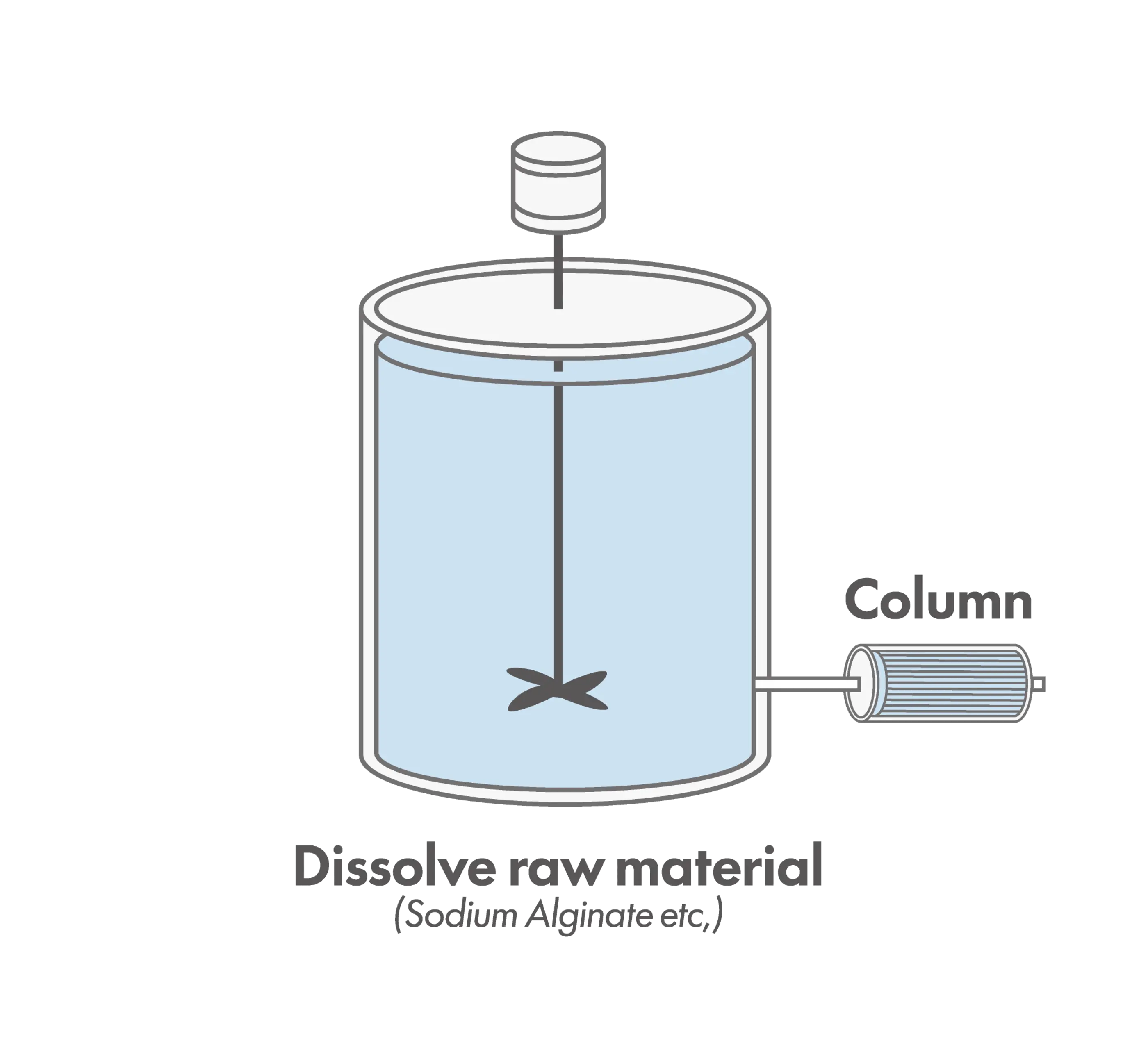
In the column-based method, adsorption media are packed into a column, and liquid samples are continuously passed through to remove endotoxins. Because flow rate and contact time can be precisely controlled, this approach offers high reproducibility and is well-suited for manufacturing processes and continuous operations. However, it requires appropriate equipment design and involves higher initial costs.
Column-based methods can be further classified according to their adsorption principles, as outlined below.
| Comparison Item | Standard Column | Syringe Column | Spin Column |
|---|---|---|---|
| Operating principle | Gravity flow or pump-driven flow | Manual pressure | Centrifugal force |
| Processing scale | Several mL to several hundred liters Suitable for manufacturing processes | 1 mL to several mL Suitable for simple, small-scale processing | 10 µL to 20 mL Suitable for laboratory screening |
| Contact time control | The flow rate can be adjusted to ensure sufficient binding time | Varies depending on manual operation; slow and careful operation is required | Dependent on centrifugal force and resin density; insufficient contact occurs if the sample passes through too quickly |
| Typical applications | Final product manufacturing Process development Purification of large-volume samples | On-site or field-based simple processing Use in environments without dedicated equipment | Screening of multiple samples Small-scale experiments Processing of valuable, limited-volume samples |
Batch Method
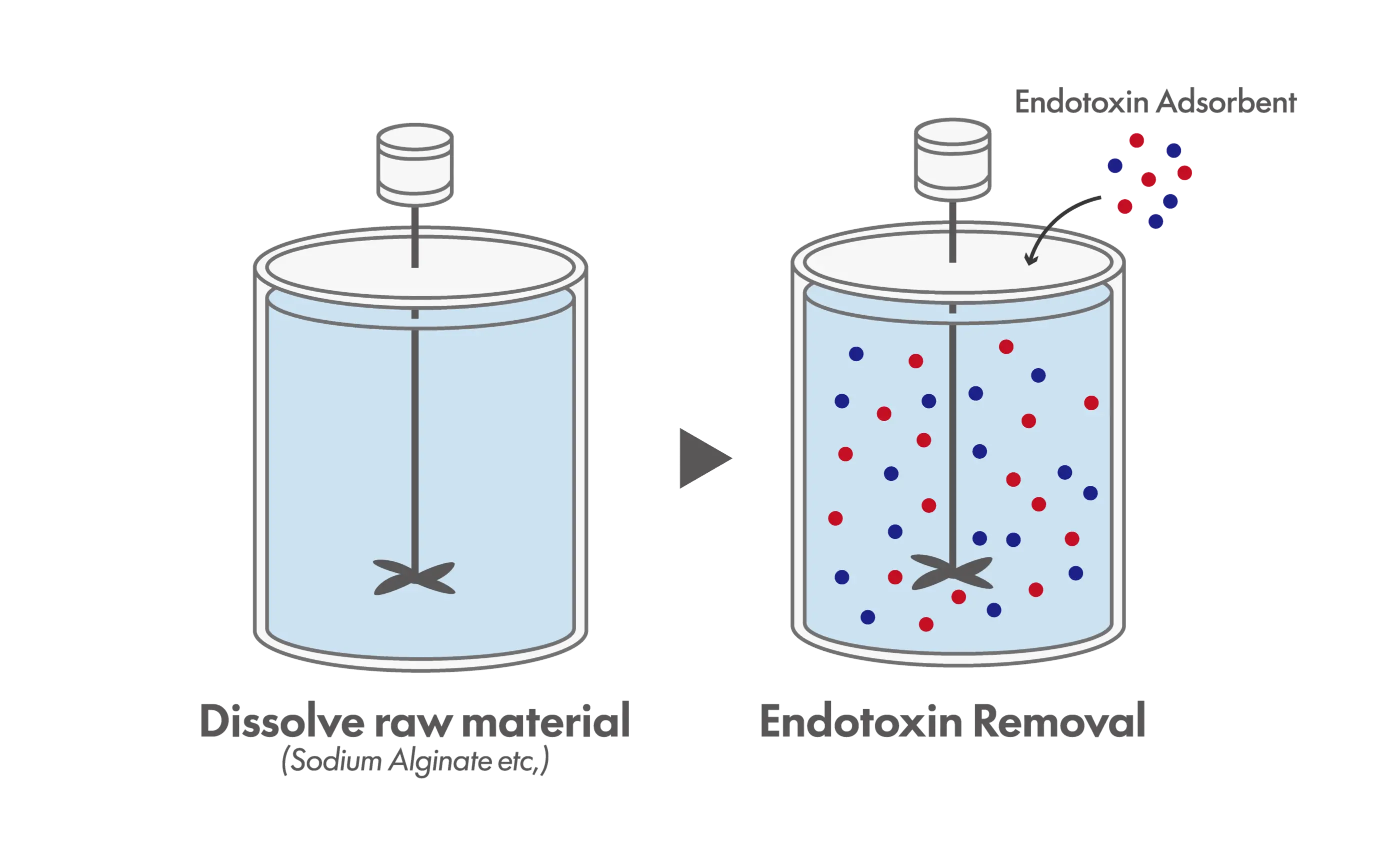
In the batch method, adsorbent materials such as beads or resins are directly added to the process solution and incubated with stirring for a defined period to allow endotoxin adsorption. The adsorbents are then removed by centrifugation or filtration. Although the theoretical removal efficiency may be lower than that of column-based methods, a key advantage is the ability to process high-viscosity solutions or slurry-like samples that cannot be readily packed into columns. In recent years, batch processing using magnetic beads, which offer simple and convenient handling, has also become increasingly common.
Nagase ChemteX’s Endotoxin Removal Technology
Conventional column-based methods and general-purpose adsorbents have faced challenges when processing high-viscosity materials such as sodium alginate and xanthan gum. Attempting to pass these materials through columns often results in clogging or leads to degradation of the adsorbent media and columns due to high pressure.
To address these challenges, Nagase ChemteX has established a proprietary endotoxin removal technology.
Proprietary Adsorbents with High Selectivity and High Adsorption Capacity
Nagase ChemteX’s endotoxin adsorbents were developed in collaboration with Kumamoto University. The carrier materials and ligands have been specifically designed to selectively adsorb endotoxins with higher specificity than conventional endotoxin adsorbents.
These adsorbents also exhibit high endotoxin adsorption capacity, enabling efficient removal even from materials with high initial endotoxin levels. Based on this technology, Nagase ChemteX provides solutions in three different formats, tailored to specific development phases and application needs.
Endotoxin Removal Columns (For Research and Testing Use)
For laboratory-scale purification of small-volume samples, the cartridge-type column “NEA-1000R” is recommended. It features high selectivity, enabling the removal of endotoxins while preserving valuable components such as proteins.
- High recovery with excellent selectivity
In protein solutions such as bovine serum albumin (BSA) and γ-globulin, endotoxin removal rates of ≥99% have been achieved, while maintaining protein recovery rates of ≥95%. This high selectivity helps minimize the loss of valuable samples.
- Simple operation (Ready-to-use)
The product is supplied as individually packaged, ethylene oxide gas (EOG)–sterilized units. It can be used immediately by connecting to a syringe or peristaltic pump and rinsing with WFI or a suitable equivalent. No special preparation or complex setup is required.
- Broad applicability
In addition to protein solutions, the column is suitable for processing a wide range of samples, including culture media, nucleic acids, and polysaccharides such as pullulan.
| Sample | Isoelectric Point (pI) | Before Treatment | After Treatment | Removal of Endotoxin Rate | Protein Recovery Rate |
|---|---|---|---|---|---|
| BSA | 4.9 | 30 EU/mL | 0.1 EU/mL | ≧99% | ≧98% |
| γ-globulin | 7.4 | 41 EU/mL | 0.5 EU/mL | ≧99% | ≧99% |
| Cytochrome C | 10.6 | 27 EU/mL | 0.1 EU/mL | ≧99% | ≧96% |
Low-Endotoxin Biomaterials Arcofeliz™
In the development of regenerative medicine products and medical devices, natural polymers such as sodium alginate and xanthan gum have attracted significant attention due to their high biocompatibility. However, because these materials are derived from natural sources, they present major challenges, including high levels of impurities such as endotoxins and high viscosity, which makes purification particularly difficult.
The Arcofeliz™ series from Nagase ChemteX is a line of high-purity, low-endotoxin biomaterials developed using the company’s proprietary removal technology to address these challenges. These materials are manufactured with Nagase ChemteX’s unique endotoxin removal technology, enabling endotoxin levels to be reduced to ranges suitable for medical applications without compromising the intrinsic viscosity or physical properties of the materials.
Product Lineup
Multiple product variants are available to support a wide range of applications, including scaffolds for regenerative medicine, hemostatic materials, anti-adhesion materials, and drug delivery system (DDS) matrices.
- Sodium Alginate
A polysaccharide derived from seaweed.
It is suitable for applications such as cell encapsulation and wound dressings.
Nagase ChemteX’s proprietary endotoxin removal technology enables endotoxin reduction while preserving the intrinsic physical properties of the material, allowing for a broad lineup with varying specifications.
・M/G ratio: M/G≧1.5, M/G≦0.8, M/G≒1.0
・Viscosity: 1–1,200 mPa·s
- Xanthan Gum
A polysaccharide produced by Xanthomonas campestris, a Gram-negative bacterium.
It exhibits high thickening performance and is widely used as a pharmaceutical excipient and as a raw material for functional gels.
Because it is derived from Gram-negative bacteria, xanthan gum typically contains high levels of endotoxins and is difficult to process into low-endotoxin grades. Nagase ChemteX’s technology makes it possible to achieve low endotoxin levels while maintaining high viscosity.
- Gelatin
An animal-derived protein.
It is widely used as a material for cell culture substrates and implantable devices.
Due to the risk of zoonotic contamination, the supply of low-endotoxin gelatin can be unstable. Nagase ChemteX enables the stable production and supply of low-endotoxin gelatin with reduced dependence on raw material lot variability.
- Others
Additional biomaterials are also available in the product lineup.
For more details, please contact us.
Contract Services: Endotoxin Removal Evaluation and Processing
This contract service involves receiving customer-owned materials and evaluating optimal conditions for endotoxin removal. The service supports process implementation across a wide range of scales, from feasibility studies at the milliliter level to scale-up at the liter level.
In the development of new biopharmaceuticals and regenerative medicine products, endotoxin removal from raw materials is an unavoidable challenge. However, identifying optimal removal conditions in-house and subsequently scaling the process to manufacturing levels requires substantial resources and specialized expertise.
Nagase ChemteX offers contract services that cover the entire workflow, from condition screening to contract processing for endotoxin removal, while carefully handling customers’ valuable materials. Leveraging extensive experience with a wide variety of materials, including high-viscosity substances and proteins, Nagase ChemteX proposes and establishes optimized processes for washing, endotoxin removal, and material recovery tailored to each customer’s material.
Services are available from small-scale laboratory studies through to large-scale plant production. Please feel free to contact us for further discussion.
【References】
Tsuji, K., et al. (1978). Applied and Environmental Microbiology, 36(5), 710-714.
Csako, G., et al. (1983). Applied and Environmental Microbiology, 45(4), 1342-1350.
Ludwig, J. D., & Avis, K. E. (1985). Journal of Parenteral Science and Technology, 39(6), 258-261.
Munford, R. S., et al. (2009). The Journal of Immunology, 182(10), 5901-5908.
Bates, J. M., et al. (2007). Cell Host & Microbe, 2(6), 371-382.
Aida, Y., & Pabst, M. J. (1990). Journal of Immunological Methods, 132(2), 191-195.
Petsch, D., & Anspach, F. B. (2000). Journal of Biotechnology, 76(2-3), 97-119.
Anspach, F. B. (2001). Journal of Chromatography A, 921(1), 31-46.
Guo, W., et al. (1990). Biomedical Chromatography, 4(2), 67-73.
Hirayama, C., et al. (1994). Journal of Chromatography A, 676(2), 267-275.
- 【Important Notice Regarding Pharmaceutical and Medical Device Regulations】
- Positioning of the technology: The products and technologies introduced in this article are intended for use as raw materials or processing technologies in the manufacturing processes of pharmaceuticals and medical devices, or in research and development activities. They do not guarantee the efficacy or safety of final products, such as pharmaceutical products.
- Regarding Arcofeliz™: The Arcofeliz™ series consists of materials intended for use as pharmaceutical excipients or raw materials for medical devices. These materials themselves are not pharmaceutical products intended for the diagnosis, treatment, or prevention of diseases.
- Quality specifications: Expressions such as “low endotoxin” refer to physicochemical properties based on each product’s specification values and do not claim or imply any clinical effects.
Related Products
We’re Here to Help
Complete the following fields with your information, as well as a short, detailed description of your request and a NAGASE Specialist will be in touch with you shortly.


















