Endotoxin Control in Sodium Alginate
Limitations of Low-Endotoxin Processing and Practical Solutions
April 02, 2026
Sodium alginate is a polysaccharide widely used in regenerative medicine, cell culture, and medical device applications due to its excellent biocompatibility and gel-forming ability. However, endotoxin contamination originating from raw materials and manufacturing processes can be a critical issue. In applications that directly interact with living systems, minimizing endotoxin levels is essential.
This article outlines the relationship between sodium alginate and endotoxins, explains why achieving low-endotoxin grades is technically challenging, and introduces commonly used countermeasures and their limitations.

What Is Sodium Alginate?
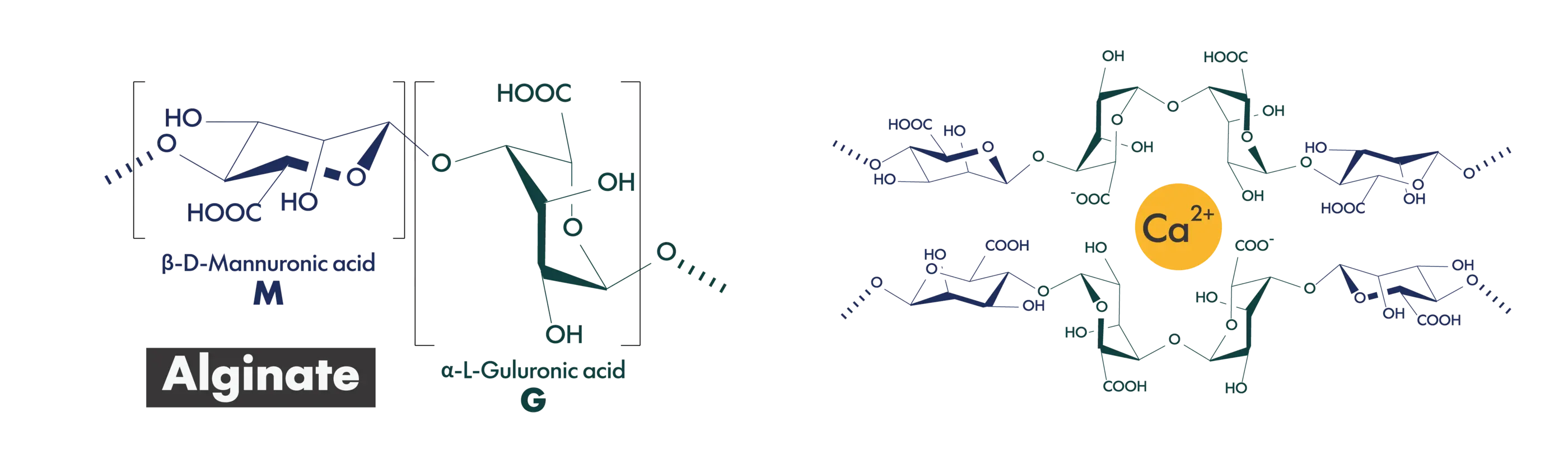
Sodium alginate is a high-molecular-weight polymer obtained by purifying alginic acid—a naturally occurring polysaccharide found in brown seaweeds such as kelp and wakame—into its sodium salt form. It is primarily composed of two uronic acid residues, mannuronic acid (M) and guluronic acid (G), and exhibits high viscosity when dissolved in water.
Sodium alginate also has the unique ability to form gels through ionic crosslinking with divalent metal ions such as calcium. Owing to this property, it is widely used as a thickening agent, stabilizer, and gelling agent in the food, pharmaceutical, and cosmetic industries. In recent years, its excellent biocompatibility has driven rapidly growing interest in its use for pharmaceutical and medical device applications, as well as in regenerative medicine and cell therapy, where it serves as a scaffold material for cell culture and as a matrix for cell encapsulation.
Effects of Sodium Alginate on Cells
Sodium alginate is widely used as a three-dimensional (3D) cell culture scaffold in regenerative medicine and tissue engineering because of its extremely low cytotoxicity and its ability to form gels under mild conditions.
The hydrogel formed through ionic crosslinking with calcium ions provides an environment that closely resembles the extracellular matrix (ECM) in vivo. Within this soft and hydrated matrix, cells can efficiently receive oxygen and nutrients while maintaining their native functions, enabling proliferation and differentiation. In particular, studies using embryonic stem (ES) cells, induced pluripotent stem (iPS) cells, and mesenchymal stem cells (MSCs) have demonstrated that cell encapsulation in alginate gels offers physical protection and a favorable microenvironment, thereby promoting spheroid formation and organoid development.
| Cell Type | Objective / Application | Role of Sodium Alginate and Outcomes |
|---|---|---|
| Human ES cells (hESCs) | Large-scale expansion Maintenance of pluripotency | Physical protection Alginate capsules act as a protective barrier, shielding cells from shear stress in bioreactors and enabling efficient cell proliferation while maintaining an undifferentiated state. |
| Mesenchymal stem cells (MSCs) | Bone regeneration Induced differentiation | Differentiation scaffold Within RGD peptide–modified alginate gels, MSCs efficiently differentiate into osteoblasts with confirmed mineral deposition. Gel stiffness significantly influences differentiation efficiency. |
| Cancer cells | Drug screening Spheroid formation | Biomimetic environment Composite gels of alginate and chitosan enable the formation of tumor-like spheroids that more closely resemble in vivo tumors. These spheroids exhibit higher drug resistance than 2D cultures, allowing more physiologically relevant drug screening. |
| Neural cells | Neural regeneration | Directional growth control Tubular alginate gels guide linear cell growth along defined channels, promoting aligned neural outgrowth. |
| Chondrocytes | Phenotype maintenance Suppression of dedifferentiation | Prevention of dedifferentiation Alginate-based 3D culture suppresses fibroblast-like dedifferentiation observed in 2D culture and maintains cartilage-specific matrix production for over eight months. |
| Pancreatic islets | Diabetes treatment Immune isolation | Immune barrier Alginate capsules function as a semipermeable membrane that allows oxygen and nutrients to pass while blocking immune cells. Successful normalization of blood glucose levels has been reported without the use of immunosuppressive drugs after transplantation. |
Effects of the M/G Ratio of Sodium Alginate on Cellular Behavior
The ratio of guluronic acid (G) to mannuronic acid (M) in sodium alginate—commonly expressed as the M/G ratio—has a profound impact on the physical properties of the resulting gel, such as stiffness and elasticity. These changes in mechanical properties directly influence cellular behavior, including cell viability, proliferation, and differentiation.
| M/G Ratio | Target Cells | Key Findings |
|---|---|---|
| G Rich (M/G ≦ 0.8) | Pancreatic islet model cells: βTC3 | Structural stability The gel exhibits minimal swelling and high mechanical stability during long-term culture. However, the dense network structure reduces nutrient diffusion efficiency. |
| M Rich (M/G ≧ 1.5) | Pancreatic islet model cells: βTC3 | Permeability and cell proliferation Enhanced mass transfer allows improved nutrient diffusion, resulting in higher cell proliferation and metabolic activity. |
| Standard (M/G ≒ 1.0) | Chondrocytes | Compared with M-Rich and G-Rich alginates, this balanced composition shows superior cell viability and phenotype preservation. Expression of cartilage-specific genes such as SOX9, COL2, and aggrecan is significantly upregulated. |
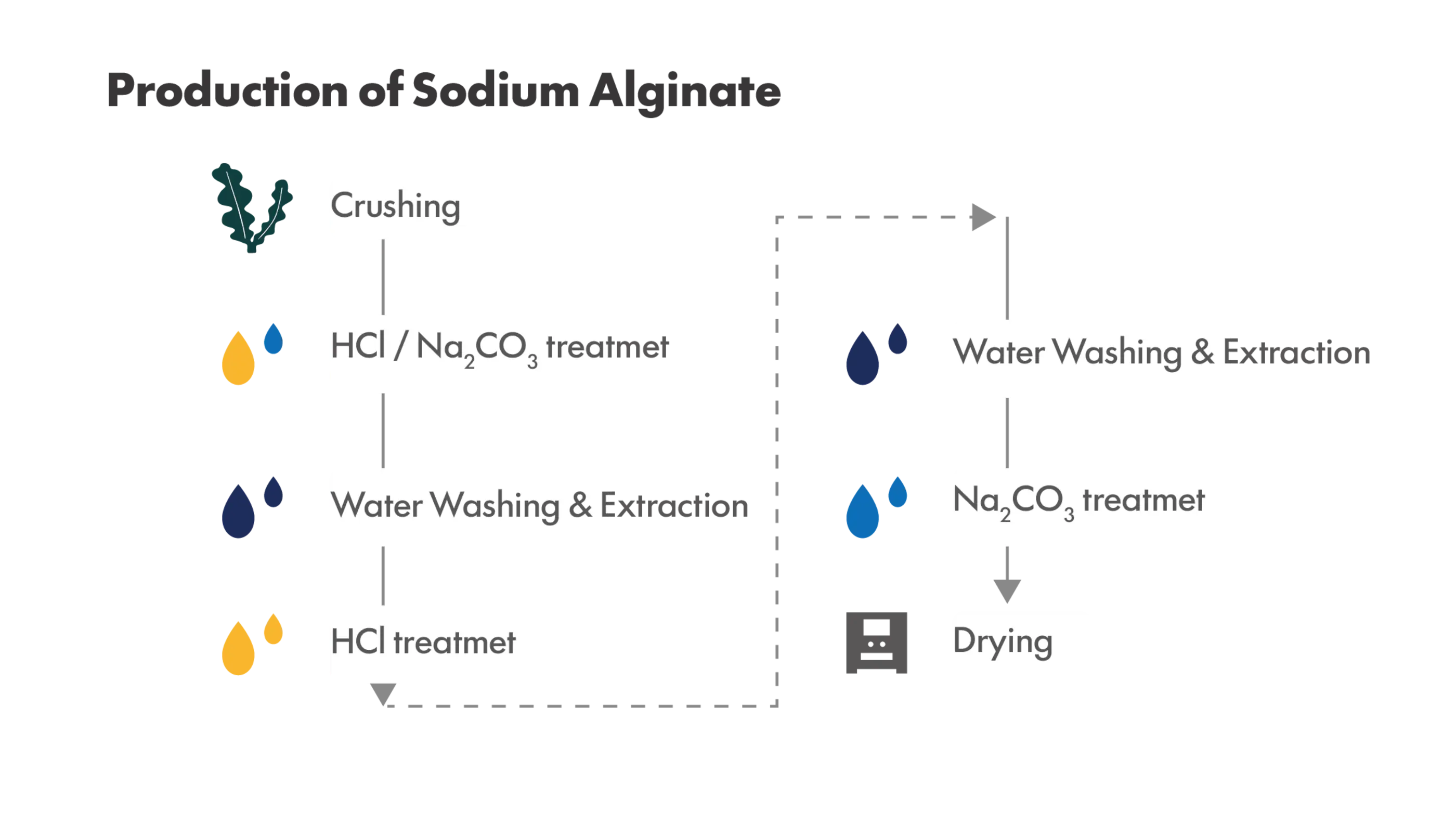
What Is Low-Endotoxin Sodium Alginate?
Low-endotoxin sodium alginate refers to sodium alginate in which endotoxins originating from raw materials and manufacturing processes have been reduced to the lowest possible levels.
While trace amounts of endotoxins may not pose a problem in general applications such as food products, they represent a significant risk in applications that directly interact with living systems, including regenerative medicine, cell culture, and medical devices. Even very low levels of endotoxins can trigger inflammatory responses in cells or interfere with differentiation processes. Therefore, for pharmaceutical, medical device, regenerative medicine, and cell therapy applications, the selection of low-endotoxin–grade sodium alginate is critically important.
Endotoxins Present in Sodium Alginate
The primary reason endotoxins are present in sodium alginate is that its raw materials are derived from natural sources such as brown seaweeds. Marine environments harbor large populations of Gram-negative bacteria, and endotoxins originating from these bacteria can adhere to or coexist with the seaweed, entering the raw material stream during harvesting.
In addition, during the extraction and purification of alginic acid, endotoxins are readily released as microorganisms are destroyed. Due to the highly viscous and complex polysaccharide structure of alginate, these endotoxins tend to become entrapped within the polymer matrix. As a result, conventional purification steps such as washing and filtration are often insufficient to remove endotoxins completely, and achieving low-endotoxin sodium alginate requires specially designed processing strategies.
Applications of Low-Endotoxin Sodium Alginate
Low-endotoxin sodium alginate is known to have a significant impact on cellular responses and biological activity. Representative examples of its effects on cell behavior reported in the literature are outlined below.
| Target Cells | Primary Effect | Description |
|---|---|---|
| Human mesenchymal stem cells (hMSCs) | Inhibition of osteogenic differentiation | The presence of endotoxins increases the secretion of inflammatory cytokines such as IL-6 and IL-8 via activation of the TLR4 receptor. As a result, alkaline phosphatase (ALP) activity and mineralization—key indicators of bone formation—are significantly suppressed. |
| Pancreatic islets | Fibrotic overgrowth | Contamination with polyphenols, endotoxins, or proteins induces inflammation at the transplantation site, leading to fibrotic encapsulation of the alginate capsules and loss of function. |
| Macrophages | Strong inflammatory response | Trace impurities such as endotoxins, peptidoglycan, and flagellin stimulate Toll-like receptors (TLRs), triggering the release of pro-inflammatory cytokines. |
| Lymphocytes / Monocytes | Mitogenic activation | Endotoxin and protein contamination abnormally activates and proliferates immune cells, resulting in severe immune rejection responses. |
As described above, low-endotoxin sodium alginate is an essential material for applications that directly interact with cells and living tissues, as it is critical for ensuring safety, preserving biological functionality, and achieving reliable and reproducible evaluation outcomes.
Challenges in Reducing Endotoxin Levels in Sodium Alginate
Although sodium alginate possesses advantageous properties such as excellent biocompatibility and gel-forming ability, it is also a high-molecular-weight polymer whose characteristics are highly sensitive to the processing conditions commonly used for endotoxin removal. In particular, heat treatment and chemical processing can be effective for reducing endotoxin levels but often compromise the intrinsic physical properties of sodium alginate.
This section outlines the major challenges encountered when attempting to reduce endotoxin levels in sodium alginate while preserving its functional performance.
| Processing Method | Principle of Endotoxin Inactivation | Impact on Sodium Alginate / Key Challenges |
|---|---|---|
| Dry heat treatment | Thermal degradation by high-temperature heating (e.g., ~250 °C) | Insufficient heat resistance Sodium alginate begins to degrade and carbonize at much lower temperatures (approximately 60–100 °C) than those required for endotoxin destruction, making this method unsuitable. |
| Oxidative treatment | Inactivation through oxidative reactions | Polymer chain scission Free radicals generated during oxidation cleave the glycosidic bonds of alginate, resulting in reduced molecular weight and viscosity. |
| Alkaline treatment | Hydrolysis under strongly alkaline conditions (pH ≥ 13) | Limited alkali resistance Before endotoxins are inactivated, sodium alginate undergoes β-elimination reactions at pH ≥ 10, leading to polymer degradation and structural alteration. |
| Membrane processing (UF/RO) | Size-based separation by filtration membranes | Difficult separation (1) High viscosity causes membrane fouling and gel-layer formation. (2) Endotoxin micelle formation results in molecular sizes overlapping with sodium alginate, preventing effective separation. |
| Adsorption treatment | Adsorption via electrostatic and hydrophobic interactions | Non-specific adsorption and high viscosity (1) Sodium alginate also carries a negative charge and is co-adsorbed with endotoxins, reducing recovery yield. (2) High viscosity limits diffusion into the adsorbent pores, resulting in insufficient endotoxin removal. |
Challenges Associated with Dry Heat Treatment
Dry heat treatment, which is widely used for sterilizing glassware and laboratory equipment, cannot be applied to sodium alginate because it induces degradation of the polymer itself.
Sodium alginate is a linear polysaccharide composed of mannuronic acid (M) and guluronic acid (G) units. When exposed to elevated temperatures, the glycosidic bonds in the polysaccharide backbone undergo hydrolytic and thermal cleavage, resulting in polymer chain scission. Consequently, the intrinsic viscosity of sodium alginate decreases significantly, and its gel-forming ability is lost.
- Endotoxin Inactivation Conditions
Endotoxins require heating at approximately 250 °C for 30 minutes or 180 °C for 3–4 hours to achieve thermal degradation.
- Thermal Stability Limit of Sodium Alginate
In contrast, sodium alginate begins to degrade at much lower temperatures, with decomposition starting at approximately 60–100 °C.
Challenges Associated with Oxidative Treatment
Oxidizing agents such as hydrogen peroxide can decompose and inactivate endotoxins. However, oxidative treatment generates free radicals (e.g., hydroxyl radicals, •OH) that also attack the polysaccharide chains of sodium alginate. These radicals cleave the glycosidic bonds, leading to a reduction in molecular weight and a significant decrease in viscosity.
As a result, oxidative treatment is not a practical approach for reducing endotoxin levels in sodium alginate when the preservation of viscosity and mechanical strength is required, such as in applications involving cell culture and tissue engineering.
Challenges Associated with Alkaline Treatment
Endotoxins can be inactivated under strongly alkaline conditions, such as treatment with 0.1–1.0 M NaOH (pH ≥ 13), where the ester bonds of the fatty acid chains in Lipid A—the toxic center of endotoxins—are hydrolyzed (saponified), resulting in loss of endotoxin activity.
In contrast, sodium alginate is highly sensitive to alkaline conditions. At pH values above approximately 10, β-elimination reactions readily occur, leading to cleavage of the glycosidic bonds in the polysaccharide backbone and a reduction in molecular weight. Furthermore, exposure to strong alkaline conditions may alter the M/G ratio or block distribution, which in turn affects calcium-mediated gel formation and the mechanical properties of the resulting hydrogel.
- Endotoxin Degradation Conditions (pH ≥ 13)
Under strongly alkaline conditions, such as treatment with 0.1–1.0 M NaOH, the ester bonds of Lipid A—the toxic core of endotoxins—are hydrolyzed (saponified), resulting in loss of endotoxin activity.
- Degradation Conditions of Sodium Alginate (pH ≥ 10)
When the pH exceeds approximately 10, sodium alginate undergoes a characteristic degradation reaction known as β-elimination. This reaction cleaves glycosidic bonds in the polysaccharide backbone, leading to a reduction in molecular weight.
Challenges Associated with Ultrafiltration (UF) and Reverse Osmosis (RO) Membrane Processing
In aqueous solutions, endotoxins exist not only as monomers but also form large aggregates (micelles) through hydrophobic interactions. As a result, they cannot pass through membranes with molecular weight cutoffs above a certain threshold. Consequently, only water and low-molecular-weight components permeate the membrane, while endotoxins are retained on the upstream side.
By exploiting the molecular size of endotoxins in their monomeric and micellar forms, ultrafiltration (UF) and reverse osmosis (RO) membranes can, in principle, be used to reduce endotoxin levels. Because these membrane processes do not require heat or chemical reagents, they may appear suitable for low-endotoxin processing of sodium alginate. However, in practice, high solution viscosity and overlapping molecular size distributions present significant challenges.
- Reduced Processability Due to High Viscosity
Sodium alginate exhibits very high viscosity in aqueous solutions, which originates from its large molecular weight and extensive entanglement of polymer chains. This high viscosity significantly reduces fluidity, leading to increased pressure losses during filtration. As a result, membrane fouling, filter clogging, and the formation of gel layers on membrane surfaces can occur, making the separation process difficult to operate and control.
- Overlap in Molecular Size Distribution
Although membrane separation based on molecular weight can, in theory, be used to remove endotoxins, the molecular weight ranges of endotoxins and sodium alginate often overlap, limiting effective separation.
- Molecular weight of endotoxins
Monomer: approximately 10,000–20,000 Da
Micelle (aggregate): ≥ 1,000,000 Da
- Molecular weight of sodium alginate
Ranges from several thousand to several million Da
Micelle-associated endotoxins behave as very large molecules with apparent molecular weights exceeding 1,000,000 Da, similar to sodium alginate itself. As a result, separation based on membrane molecular weight cutoffs becomes ineffective.
Conversely, reducing the membrane pore size to retain endotoxins also prevents sodium alginate from passing through the membrane, leading to a drastic reduction in product recovery. For these reasons, ultrafiltration (UF) and reverse osmosis (RO) membranes are generally not suitable for low-endotoxin processing of sodium alginate.
Challenges Associated with Adsorption-Based Endotoxin Removal
Adsorption methods that exploit electrostatic and hydrophobic interactions can be effective for endotoxin removal in certain systems. However, sodium alginate is an acidic polysaccharide containing a large number of carboxyl groups (–COOH), which impart a strong overall negative charge to the polymer.
- Non-Specific Adsorption Due to Negative Charge and Reduced Recovery
Positively charged adsorbents can be used to capture negatively charged endotoxins. However, because sodium alginate itself also carries a strong negative charge, it can be co-adsorbed onto positively charged adsorbents. As a result, sodium alginate is removed together with endotoxins, leading to a significant reduction in product recovery.
- Reduced Adsorption Efficiency Due to High Viscosity
For adsorption to be effective, target substances must diffuse into the internal pores of the adsorbent. However, sodium alginate solutions exhibit high viscosity, which severely restricts mass transfer within the system. Diffusion toward the adsorbent surface becomes inefficient, requiring extended contact times to reach adsorption equilibrium or preventing sufficient endotoxin removal altogether. This leads to unstable and poorly reproducible processing performance.
Methods for Reducing Endotoxin Levels in Sodium Alginate
Various approaches involving raw material pretreatment and purification process optimization have been employed to reduce endotoxin levels in sodium alginate. However, because sodium alginate is an acidic high-molecular-weight polysaccharide whose physicochemical properties and behavior closely resemble those of endotoxins, conventional processing methods often fail to achieve sufficient endotoxin reduction. This challenge is particularly pronounced for high-viscosity grades of sodium alginate.
The following section summarizes representative methods for low-endotoxin processing of sodium alginate and their key characteristics.
| Method | Principle | Remarks |
|---|---|---|
| Ultrafiltration (UF) | Separation using a filtration membrane | Dilution is required due to high viscosity. Separation may be ineffective because endotoxins can form micelles. |
| Surfactant + Ultrafiltration (UF) | Disruption of endotoxin micelles followed by membrane separation | An additional step is required to remove the surfactant after processing. |
| Adsorption (activated carbon) | Adsorption via hydrophobic interactions | Sodium alginate is also non-specifically adsorbed, resulting in poor recovery yield. |
| Adsorption (polymyxin B–immobilized resin) | Selective adsorption via specific interactions | Extremely expensive; careful management of ligand leaching risk is required. |
Ultrafiltration (UF)
High-molecular-weight polymers such as sodium alginate cannot pass through ultrafiltration membranes, allowing endotoxins and other low-molecular-weight impurities to permeate and be removed. However, because sodium alginate solutions are highly viscous, membrane fouling and clogging occur readily.
When ultrafiltration is applied, the sodium alginate solution must be significantly diluted to prevent membrane blockage, which reduces process efficiency and complicates downstream concentration steps.
Surfactant-Assisted Ultrafiltration (UF)
Depending on their aggregation state, endotoxins may form micelles that cannot be removed by ultrafiltration alone. In such cases, surfactants can be used to disrupt these aggregates, enabling endotoxin reduction through subsequent ultrafiltration.
However, this approach does not resolve the fundamental challenge of processing high-viscosity sodium alginate solutions. In addition, an extra downstream step is required to remove the surfactant, further increasing process complexity.
Adsorption Using Activated Carbon
The toxic moiety of endotoxins, Lipid A, is composed of lipid structures and therefore exhibits strong hydrophobicity. Activated carbon has a high affinity for hydrophobic substances, allowing the lipid component of endotoxins to be adsorbed and removed by passing a sodium alginate solution through an activated carbon medium.
However, sodium alginate itself is also prone to adsorption onto activated carbon, which leads to reduced product yield. In addition, the adsorption capacity for endotoxins is relatively limited, making this method unsuitable when the endotoxin content in the solution is high.
Adsorption Using Polymyxin B–Immobilized Resins
Polymyxin B is an antibiotic that exhibits highly specific binding to the Lipid A moiety of endotoxins. When polymyxin B is immobilized on beads or membranes and a sodium alginate solution is passed through, endotoxins are selectively adsorbed while sodium alginate remains in solution.
However, polymyxin B is extremely expensive, which limits the use of this method to laboratory-scale applications or to high-value medical products where cost constraints are less restrictive.
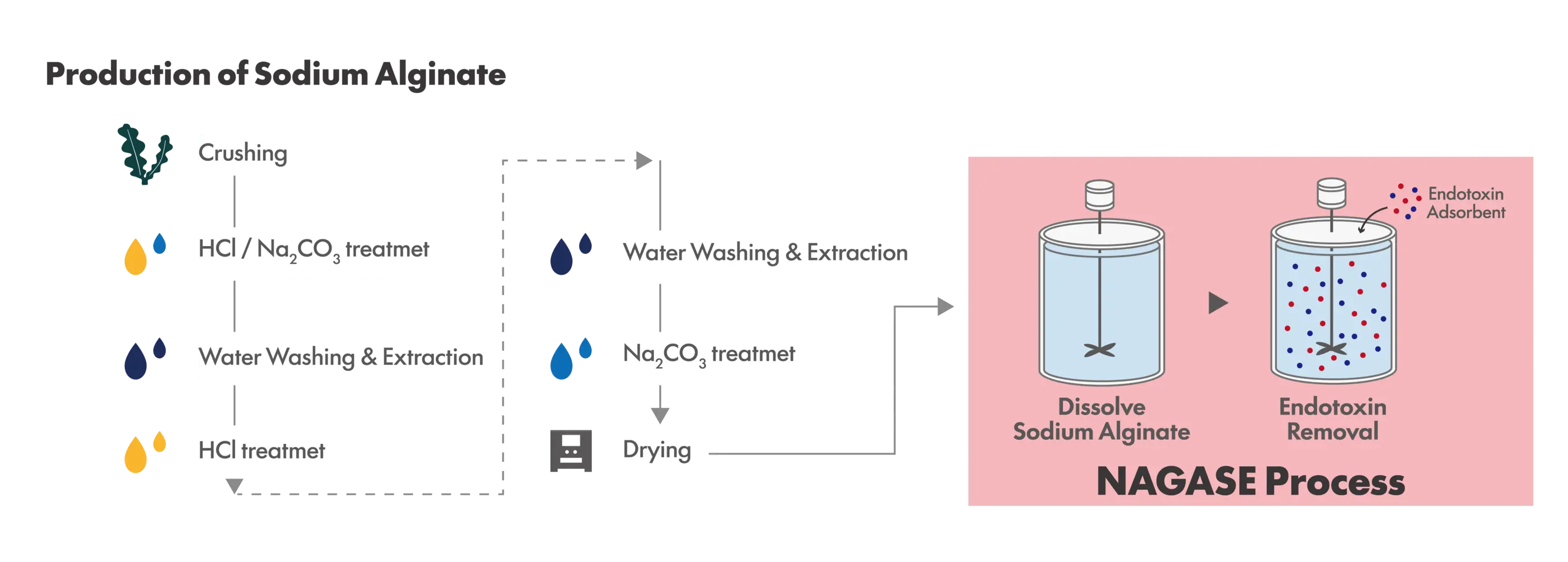
Nagase ChemteX’s Low-Endotoxin Sodium Alginate
Nagase ChemteX has developed a proprietary endotoxin adsorbent that enables selective endotoxin removal without the use of polymyxin B. By employing an adsorption-based process, endotoxins can be removed efficiently while preserving the intrinsic properties of sodium alginate.
This approach shows minimal dependence on raw material lots or compositional variations, allowing the stable production of multiple grades of low-endotoxin sodium alginate. These products are offered as the Arcofeliz™ AL series.
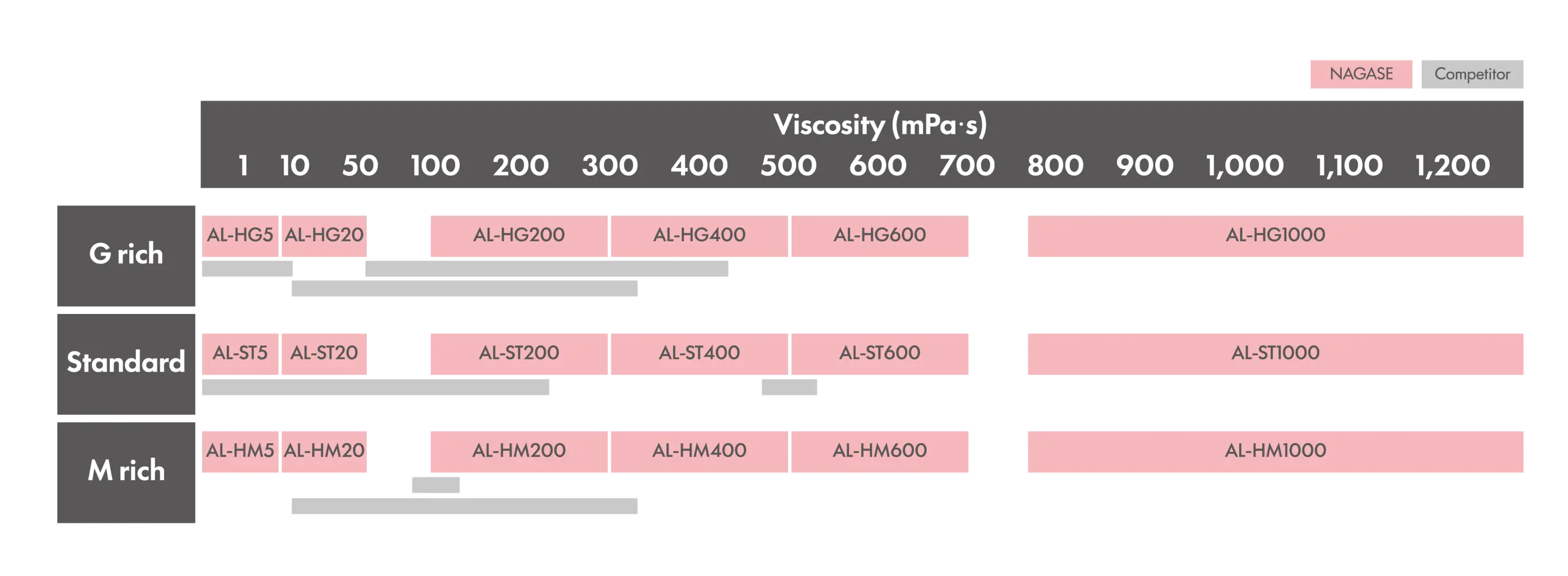
Key Features
・Strict endotoxin control: < 30 EU/g
・Wide viscosity range: 1–1,200 mPa·s
・Three M/G ratio options: M/G ≧ 1.5, M/G ≦ 0.8, and M/G ≒ 1.0
・Dedicated facilities designed specifically for low-endotoxin processing
Nagase ChemteX’s low-endotoxin sodium alginate features exceptionally low-endotoxin levels and very high purity.
| Manufacturer | Endotoxin Level |
|---|---|
| Nagase ChemteX | <10EU/g (High-purity grade) |
| <30EU/g | |
| Competitor A | <50EU/g * |
| Competitor B | <100EU/g * |
*Values are compared based on publicly available information.
If you are interested in low-endotoxin sodium alginate, please feel free to contact us for further information or consultation.
【References】
Siti-Ismail, N., et al. (2008). Biomaterials, 29(29), 3946-3952.
Wang, L., et al. (2010). Biomaterials, 31(7), 1848-1857.
Kievit, F. M., et al. (2010). Biomaterials, 31(14), 3903-3913.
Prang, P., et al. (2006). Biomaterials, 27(19), 3560-3569.
Häuselmann, H. J., et al. (1994). Journal of Cell Science, 107(Pt 1), 17-27.
Lim, F., & Sun, A. M. (1980). Science, 210(4472), 908-910.
Stabler, C., et al. (2001). Biomaterials, 22(10), 1301-1310.
Lee, B. B., et al. (2012). Journal of Biomaterials Science, Polymer Edition, 23(1-4), 185-199.
Klöck, G., et al. (1997). Biomaterials, 18(10), 707-713.
Dusseault, J., et al. (2006). Journal of Biomedical Materials Research Part A, 76(2), 243-251.
Paredes Juarez, G. A., et al. (2014). Journal of Controlled Release, 172(3), 983-992.
Paredes Juarez, G. A., et al. (2010). Journal of Biomedical Materials Research Part B, 93(2), 333-340.
Holme, H. K., et al. (2008). Carbohydrate Polymers, 73(4), 656-664.
- 【Important Notice Regarding the Pharmaceuticals and Medical Devices Act (PMD Act, formerly the Pharmaceutical Affairs Act)】
- Positioning of the technology: The products and technologies introduced in this article are intended for use as raw materials or processing technologies in pharmaceutical and medical device manufacturing processes, as well as in research and development. They do not guarantee the efficacy or safety of final products (e.g., pharmaceuticals or medical devices).
- Arcofeliz™: The Arcofeliz™ series is a material designed for use as a pharmaceutical excipient or medical device raw material. It is not a pharmaceutical product intended for the diagnosis, treatment, or prevention of diseases.
- Quality specifications: Descriptions such as “low-endotoxin” refer to physicochemical properties based on each product’s specifications and do not claim or imply any clinical effects.
Related Products
We’re Here to Help
Complete the following fields with your information, as well as a short, detailed description of your request and a NAGASE Specialist will be in touch with you shortly.


















