Peptide / Oligonucleotide Manufacturing Challenges and our Innovative Solutions
March 17, 2026
1. Challenges in Peptide and Oligonucleotide Manufacturing
Manufacturing peptides and oligonucleotides presents several critical challenges that impact cost, quality, and environmental sustainability.
- High Raw Material Costs
Solid-phase synthesis requires large excesses of protected monomers and reagents, significantly increasing raw material expenses.
- High Processing Costs
Both upstream and downstream processes involve long processing times, resulting in elevated manufacturing costs.
- Difficulty in Quality Control
Because solid-phase synthesis is performed on a solid support (resin), monitoring the progress of coupling reactions qualitatively is inherently challenging.
- Environmental Impact
Manufacturing requires large volumes of organic solvents such as DMF and MeCN, raising concerns related to environmental burden and sustainability.
2. Manufacturing Solutions
Our partner, PeptiStar, addresses these challenges through advanced technologies and deep process expertise, supported by a Japanese reliability-focused technological approach.
- Reduction of in‑process testing to a single step, improving efficiency and reliability
- Continuous chromatography that maximizes purity and yield
- Specialized expertise in peptides, oligonucleotides, and PDC manufacturing and purification
- Cost‑reduction and lead‑time shortening strategies based on extensive process knowledge.
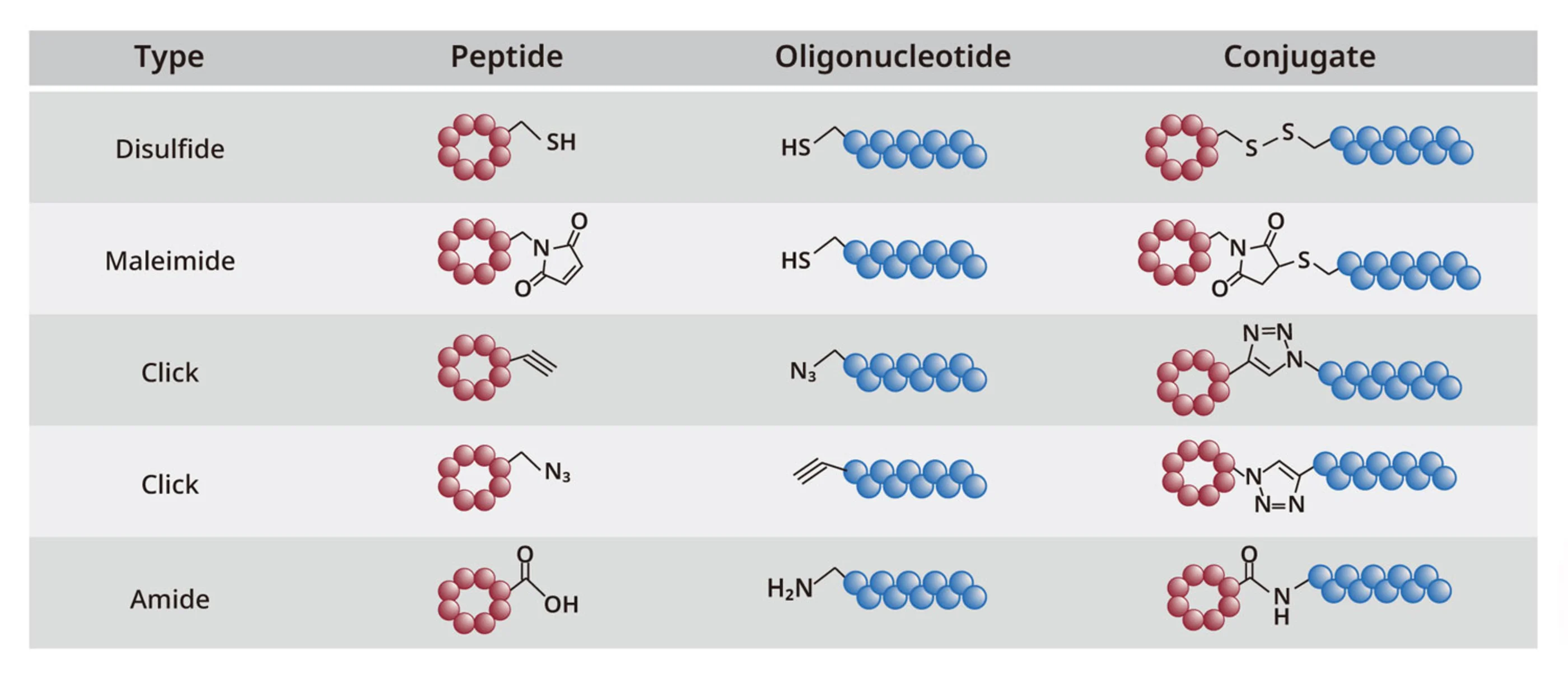
For more details on our peptide-drug conjugate (PDC) development capabilities—covering linker design, conjugation strategies, and purification approaches—please refer to our PDC technical white paper.
3. Twin-Column Continuous Chromatography
PeptiStar’s twin‑column continuous chromatography system delivers high‑purity APIs with excellent yields, enabling significant cost reductions.
- Achieves 30–50% reduction in overall cost including purification cost and raw material cost.
- Achieves the optimal balance of high yield and high purity, often unattainable with traditional batch methods.
For more detailed data, please contact us.
4. Mixer-Type Lyophilization System
PeptiStar‘s innovative mixer‑type lyophilization technology offers a breakthrough in drying efficiency and product uniformity.
- Achieves an over 80% reduction in drying time
- Provides improved homogeneity compared with conventional lyophilization.
- Enables shorter manufacturing timelines and enhanced product consistency.
For additional technical data, please reach out to us.

5. Benefits of Partnering with NAGASE and PeptiStar (CDMO Advantage)
Partnering with NAGASE and PeptiStar gives you access to a unique combination of global sourcing power, advanced technologies, and world‑class CDMO capabilities—accelerating your development while ensuring quality, reliability, and supply‑chain strength.
NAGASE brings more than 70 years of experience as a global solution provider in the pharmaceutical and healthcare industry. With a robust worldwide network, deep expertise in raw materials and functional ingredients, and a strong commitment to regulatory compliance and quality assurance, NAGASE ensures stable sourcing, risk‑managed supply chains, and seamless support from early development through commercial scale.
PeptiStar, as a cutting‑edge CDMO specializing in peptides, oligonucleotides, and PDCs, delivers manufacturing excellence through proprietary continuous chromatography, advanced lyophilization technology, and comprehensive analytical platforms. Our expertise enables high purity, superior yield, and efficient cost structures—helping you shorten timelines without compromising quality.
Together, NAGASE and PeptiStar provide end-to-end value:
- Integrated sourcing and manufacturing capabilities that enhance supply reliability and drive substantial cost and lead‑time optimization.
- Seamless technical and regulatory support backed by NAGASE’s global pharmaceutical knowledge base.
- Consistent high‑quality production powered by PeptiStar’s proprietary purification and analytical technologies.
By leveraging NAGASE’s global reach and PeptiStar’s specialized CDMO capabilities, customers gain a strategic partner that supports development, ensures operational stability, and accelerates commercialization with confidence.
Related Resources
We’re Here to Help
Complete the following fields with your information, as well as a short, detailed description of your request and a NAGASE Specialist will be in touch with you shortly.















