Simultaneous Optimization of Purity and Yield via Integrated Process Control Technologies
April 24, 2026
1. The Technical Foundations Behind PeptiStar’s Optimization Strength
As the global market for mid‑sized molecule therapeutics continues to grow, peptide drug discovery has become an increasingly important field. PeptiDream has emerged as one of the world’s leaders in this space, recognized internationally for its innovative macrocyclic peptide technology and strong global partnerships. To translate these discoveries into scalable, high‑quality peptide APIs, however, industry‑wide manufacturing bottlenecks remained. PeptiStar was established on September 1, 2017, by PeptiDream, Shionogi, and Sekisui Chemical precisely to address this challenge and serve as the strategic CDMO enabling the practical implementation of next‑generation peptide therapeutics.
Partnering with NAGASE and PeptiStar gives you access to a unique combination of global sourcing power, advanced technologies, and world-class CDMO capabilities—accelerating your development while ensuring quality, reliability, and supply‑chain strength.
2. Overview of PeptiStar’s Manufacturing Capabilities
PeptiStar offers fully integrated GMP manufacturing capabilities specializing in peptides, oligonucleotides, and peptide–drug conjugates (PDCs). Our platform combines automated synthesis, advanced purification technologies, and a comprehensive analytical infrastructure to support high‑quality development from early stage to commercial scale.
Key strengths include:
- Expertise in complex sequences, modifications, and conjugation chemistry
- Capability to oversee high‑potency APIs and multi‑step PDC production.
- Quality systems aligned with global regulatory expectations.
- Advanced analytical methods for impurity profiling and structural confirmation
3. Experiences for Unique Synthesis
PeptiStar produces long-chain peptides (over 100 residues), cyclic peptides, branched peptides, dimeric peptides, and other modified peptides. Additionally, we manufacture oligonucleotides in-house and have a proven record of creating peptide-oligonucleotide conjugates with various binding patterns.
With years of accumulated know-how and dedicated technical specialists, we can propose synthesis strategies and process optimization even for complex structures and novel molecular concepts beyond those shown here. Please contact us for further details.
| Peptide | Linear Peptide |
| Cyclic Peptide (Disulfide Bond, Thioether Bond, Amide Bond, Bicyclic) | |
| Cyclic Homo-dimer Peptide | |
| Cyclic Hetero-dimer Peptide | |
| Cyclic Tetramer Peptide | |
| Branched Peptide | |
| Long Chain Peptide (>100mer) | |
| Oligonucleotide | gRNA (>130mer) |
| DNA (ASO, Aptamer, CpG Oligo) | |
| RNA (ASO, Aptamer) | |
| siRNA | |
| PMO | |
| PNA | |
| LNA | |
| All PS form | |
| Conjugation | Peptide-Oligonucleotide Conjugation (Bond type example: Disulfide, Maleimide, Click chemistry, Amide) |
| Peptide Drug Conjugation | |
| Oligonucleotide Drug Conjugation | |
| PEG, fatty acid and spacer |
4. Twin-Column Continuous Chromatography
PeptiStar's continuous chromatography process begins with the development of single-column purification conditions, followed by the development and optimization of continuous chromatography conditions. This approach seamlessly scales up, improving recovery yield while maintaining high purity and reducing crude input. PeptiStar applies this superior continuous purification method to both peptide and oligonucleotide API manufacturing, including GMP operations.
※Twin‑Column Continuous Chromatography is a proprietary continuous purification technology developed by YMC CO., LTD.
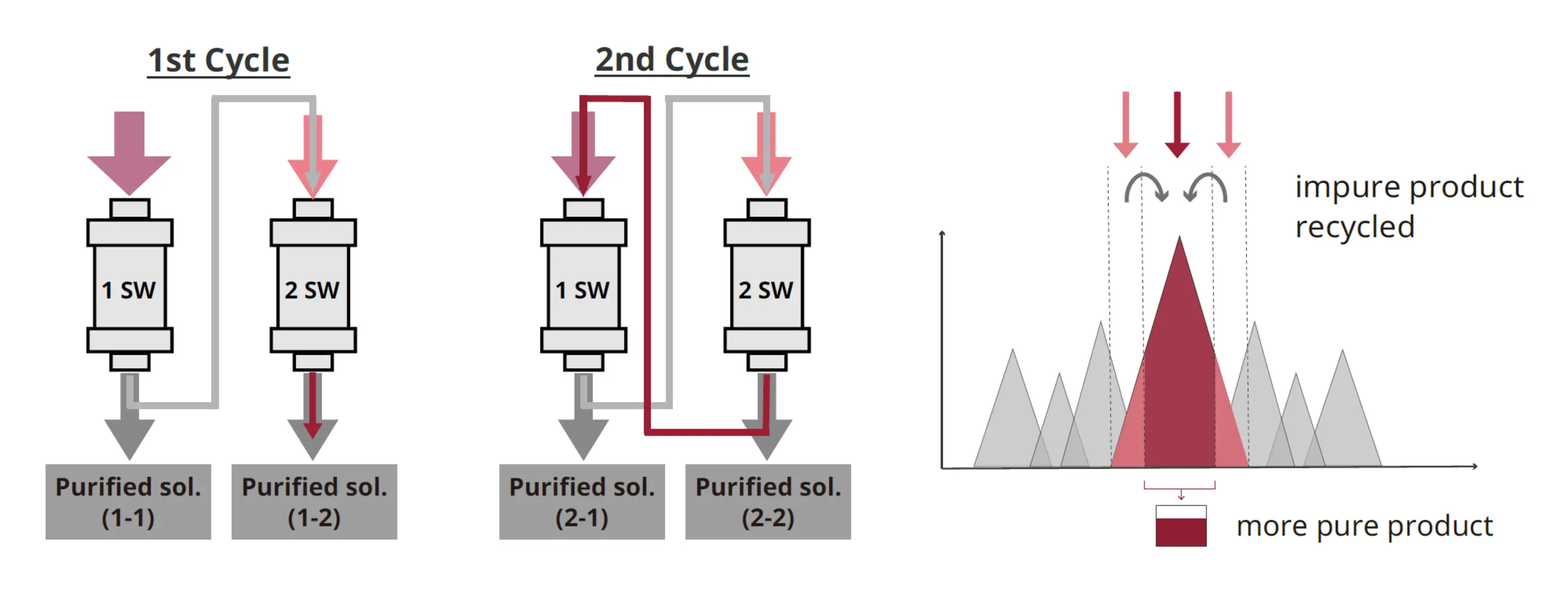
Fig. 1. Twin‑Column Continuous Chromatography
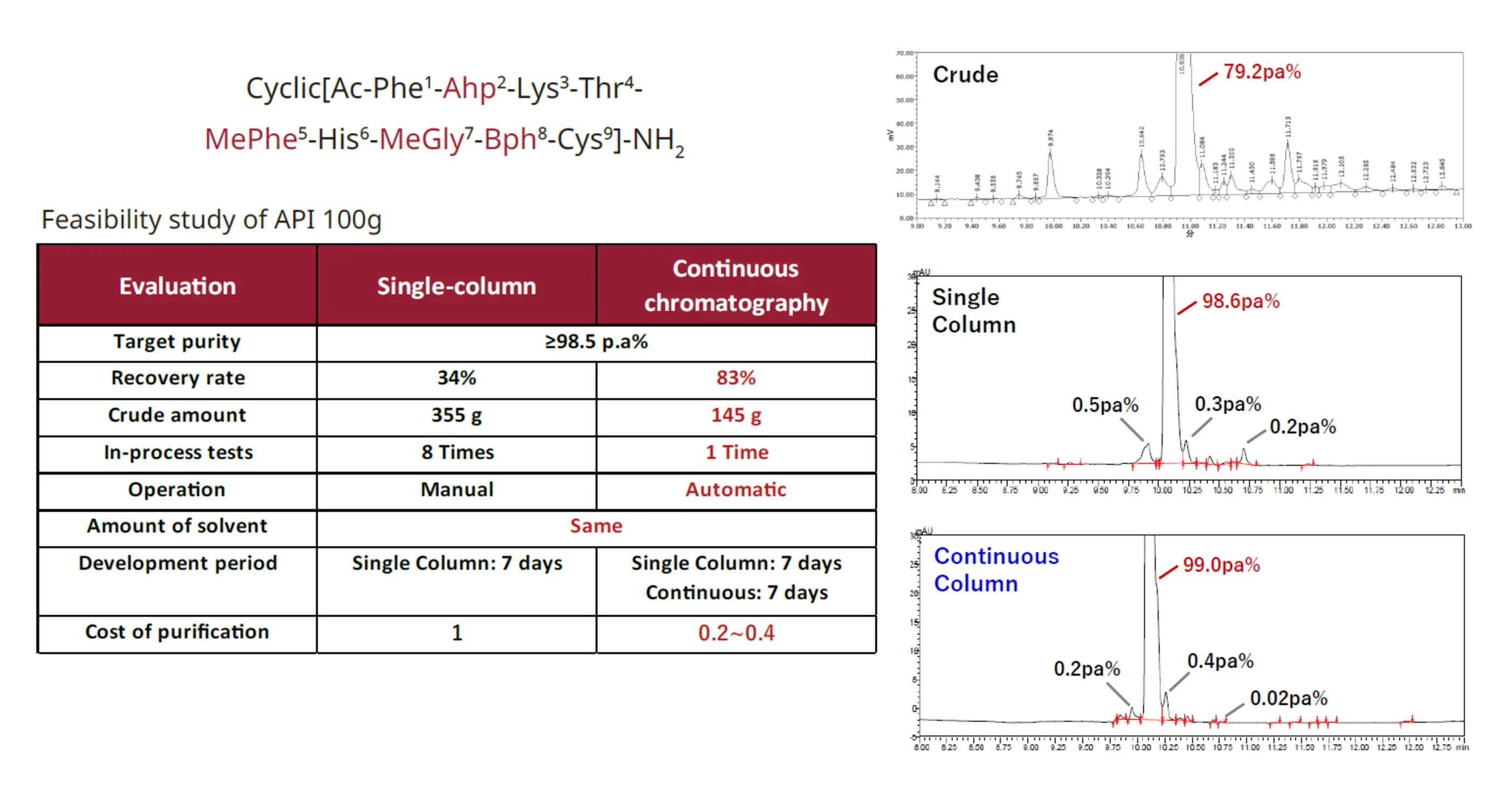
Fig. 2. Example for continuous column record (Peptide)
5. Mixer-Type Lyophilization System
Their innovative mixer‑type lyophilization technology provides a breakthrough in both drying efficiency and product uniformity. By enabling gentle yet highly effective mixing during the freezing and drying phases, this system significantly improves performance compared with conventional lyophilization.
Lyophilization studies were performed on multiple peptide compounds. As results shown in Fig.3 demonstrate that the mixer-type lyophilization system—referred to as RHEOFREED—reduced drying time by 70-80%, significantly accelerating overall manufacturing throughput.
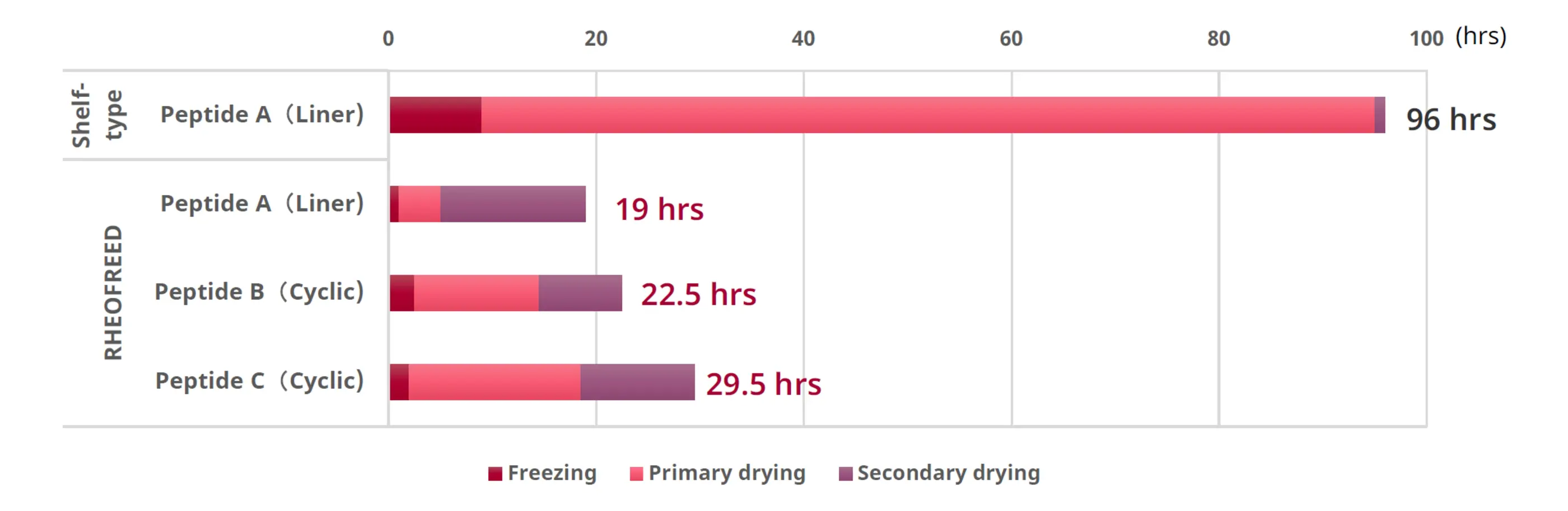
Fig. 3. Lyophilization time for various peptide compounds (RHEOFREED: Mixer-type)
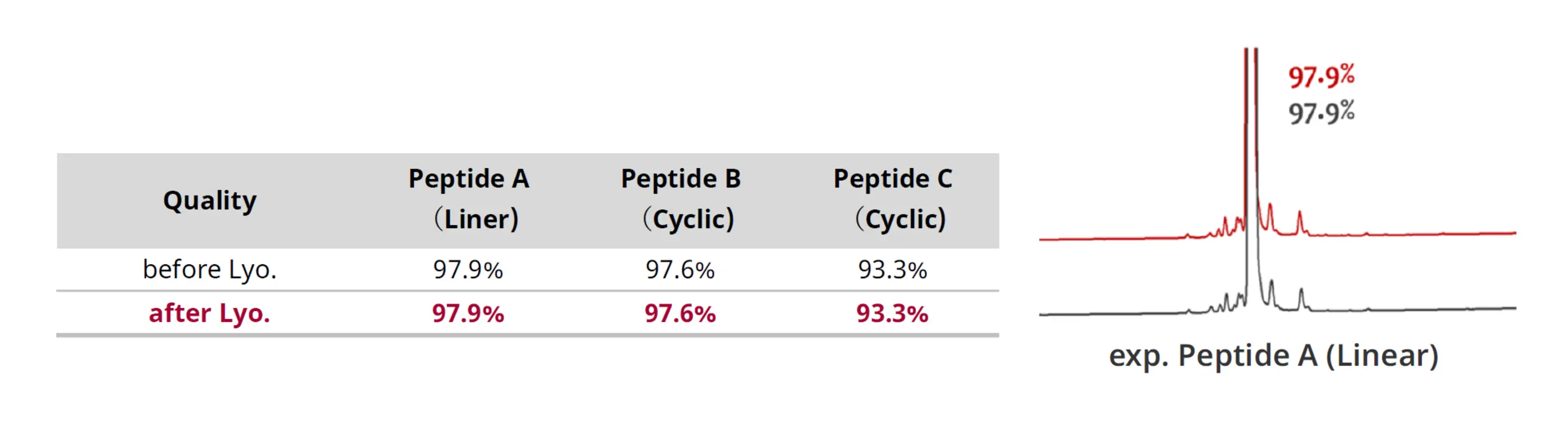
Fig. 4. Analytical data for impurities before and after lyophilization
Impurity analysis before and after lyophilization confirmed that impurity levels remained unchanged despite the shorter drying time (Fig. 4), demonstrating that product purity was maintained.
Comparative manufacturing results with conventional shelf-type lyophilization are shown in Fig. 5 and Fig. 6. Residual acetonitrile (MeCN) was significantly reduced, and evaluation of water content as an indicator of uniformity confirmed high product homogeneity
For bulk density data or additional technical information, please contact us.
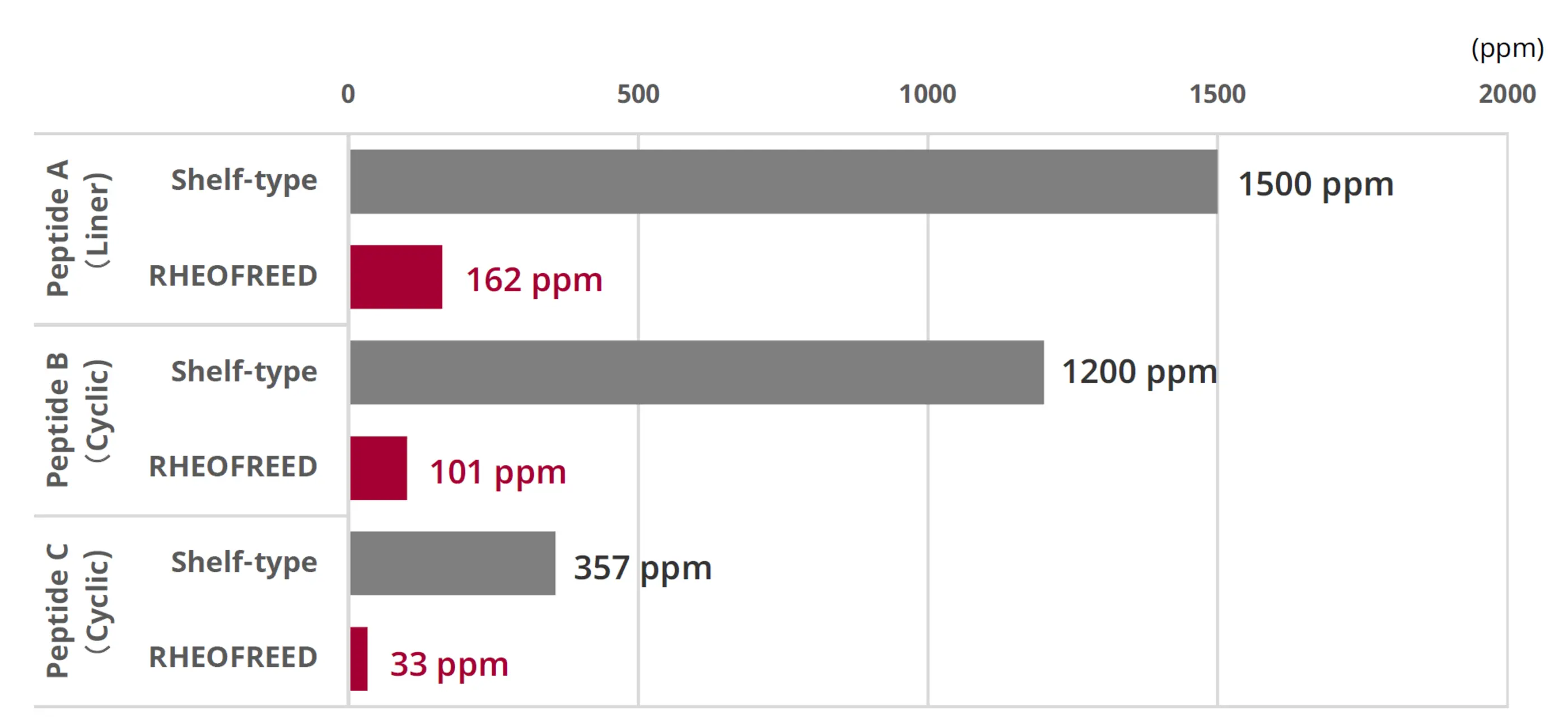
Fig. 5. Residual solvent levels based on MeCN content (RHEOFREED: Mixer-type)
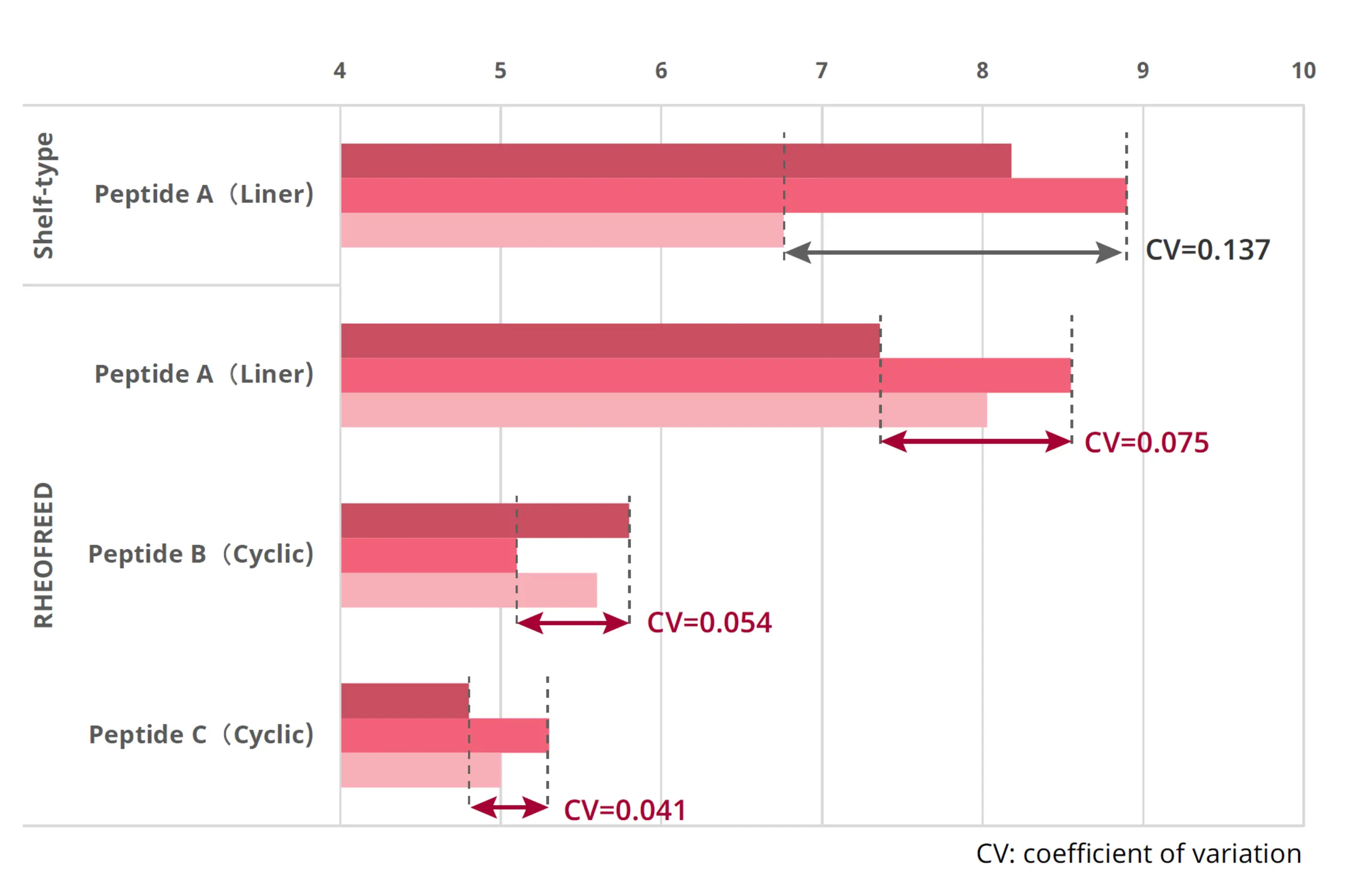
Fig. 6. Product homogeneity based on water content (RHEOFREED: Mixer-type)
6. A Leading Japanese Site for Innovative R&D and GMP Production
PeptiStar operates one of Japan’s leading R&D and GMP manufacturing sites in Settsu, Osaka. Their 10,500 m² facility integrates development, medium‑scale synthesis, and clinical‑to-commercial production, supported by advanced equipment and a team of highly skilled pharmaceutical experts.

7. Summary
PeptiStar’s advanced technologies—continuous chromatography, mixer‑type lyophilization, and a specialized analytical platform—provide measurable improvements in yield, purity, efficiency, and scalability. Combined with the global supply‑chain strength of NAGASE, we support customers with an end‑to-end solution from early development through commercial manufacturing.
Related Resources
We’re Here to Help
Complete the following fields with your information, as well as a short, detailed description of your request and a NAGASE Specialist will be in touch with you shortly.















