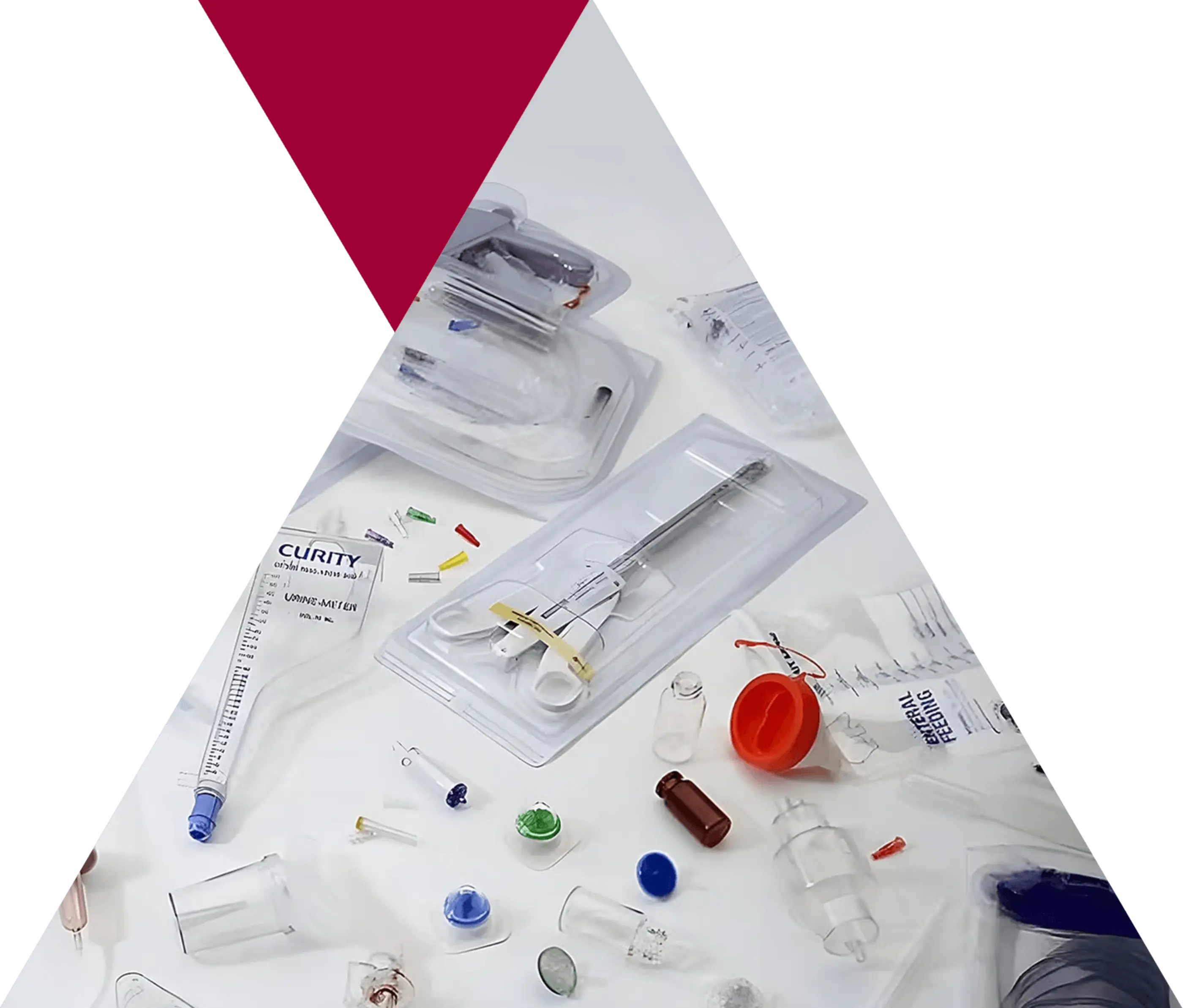
Medical Solutions Reimagined
Eastman Tritan™ Copolyester: The superior choice for safety, clarity, and durability in next-gen medical devices.
Advancing the Future of Medical Packaging in Asia
Eastman, headquartered in the USA, is a global leader in advanced materials, partnering with businesses in Thailand, Malaysia, the Philippines, and Vietnam to accelerate innovation and bring high-quality products to market faster. With over a century of expertise, Tritan™, a premium copolyester engineered for performance, safety, and design flexibility, has become the trusted choice for leading medical solutions brands seeking durable solutions that combine superior design, environmental responsibility, and compliance with international standards. This premium copolyester offers glass-like clarity, exceptional strength, and proven safety.
With a partnership spanning over 50 years, Nagase & Co., Ltd. has proudly represented Eastman in Southeast Asia—delivering the regional expertise and technical support customers rely on for success. Our long-standing collaboration allows us to provide tailored solutions, deep product knowledge, and innovative approaches that address each customer’s unique challenges.
The Challenge & The Solution
The Industry Challenge
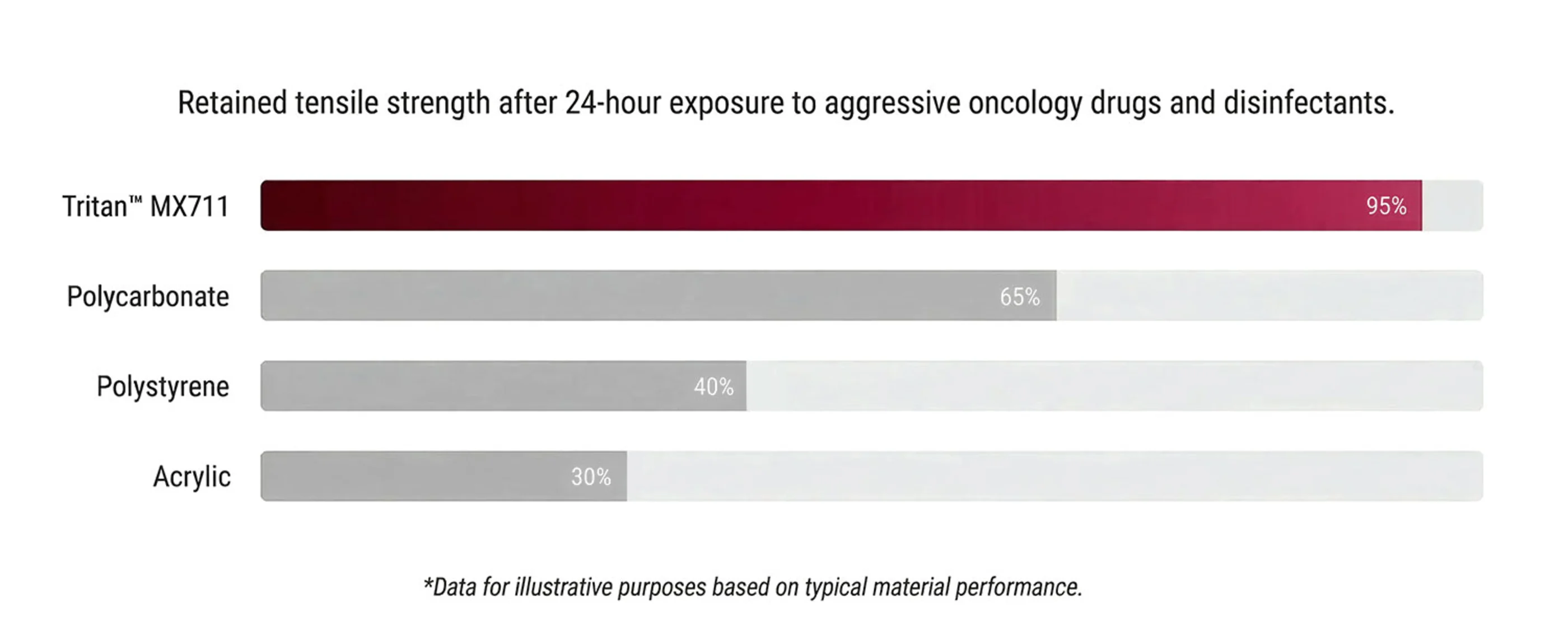
Medical environments are demanding. Aggressive disinfectants often cause traditional plastics like polycarbonate (PC) andpolystyrene (PS) to crack, haze, or fail prematurely. This compromises patient safety and increases replacement costs.
The Tritan™ Advantage
Eastman Tritan™ Copolyester is engineered to withstand the harshest chemical environments while maintaining glass-like clarity. It offers a unique balance of properties that solves the "stay clear vs.stay tough" dilemma.
Why Choose Tritan™?
Resists degradation from lipids, oncology drugs, and aggressive hospital disinfectants.
High impact resistance reduces breakage during use, transport, and manufacturing.
Free of BPA and BPS. ISO 10993 compliant for a wide range of medical applications.
Lasting glass-like transparency allows for easy visual monitoring of fluids.
Medical Applications
Fluid Management
Stopcocks, connectors, and manifolds that require clarity and chemical resistance.

Blood Therapy
Dialyzer housings and blood separation bowls that need biocompatibility and toughness.
Medical Devices
Handles and single-use devices that must withstand sterilization and impact.
Superior Chemical Resistance
| Material | Control (joules) | MCT oila | Etoposide carrier solventb | Busulfex carrier solventc | Dimethylacetamide | Dimethyl Sulfoxide |
|---|---|---|---|---|---|---|
| Tritan MX711 (standard) | 4.4 | 68 ± 13 | 90 ± 2 | 79 ± 6 | 63 ± 35 | 84 ± 2 |
| Tritan MX731 (high flow) | 4.3 | 33 ± 2 | 78 ± 23 | 39 ± 8 | 25 ± 15 | 60 ± 7 |
| PC (high flow) | 5.3 | 7 d | All broke on jig | All broke on jig | All broke on jig | All broke on jig |
| PC (Standard) | 5.5 | 34e | 12 ± 1 | All broke on jig | All broke on jig | All broke on jig |
| PC (lipid resistant) | 5.5 | 47 ± 52 | 28 ± 42 | All broke on jig | All broke on jig | All broke on jig |
| Impact-modified styrenic | 4.3 | 10 ± 1 | 7e | 8 ± 1f | Severe surface attack | 9e |
a MCT oil medium-chain triglycerides oil
b Etoposide carrier solvent: 10 mL of the solvent mix contains 3.05 mL ethanol, 6.5 of polyethylene glycol 300, 08 g polysorbate 80, 0.33 g benzyl alcohol, and 20 mg citric acid.
c Busulfex carrier solvent: 10 mL of the solvent mix contains 3.3 mL dimethyacetamide and 6.7 mL polyethylene glycol 400.
d Three of four samples broke on the jig. Standard deviation not calculated.
e Two of four samples brok on the jig. Standard deviation not calculated.
f One of four samples broke on the jig.
Safety & Sustainability

Designed for Patient Safety
Eastman Tritan™ is not manufactured with bisphenol A (BPA) or bisphenol S (BPS). It is free of estrogenic and androgenic activity (EA/AA free).
Regulatory Compliance
Supported by extensive ISO 10993 biocompatibility testing and Master Files (MAF) at the FDA, Tritan™ simplifies your regulatory pathway for 510(k) submissions.
See Tritan™ in Action Across Industries
Product Catalog and Datasheets
Eastman Tritan™ is an amorphous copolyester with excellent appearance and clarity. Its most outstanding features are excellent toughness, hydrolytic stability, and heat and chemical resistance. This new-generation copolyester can also be molded into various applications with incorporating high levels of residual stress.

Eastman Tritan™ MX711
An amorphous product with outstanding heat and chemical resistance and containing a mold release derived from vegetable based sources.
- Formulated for medical devices
- Offers excellent toughness and hydrolytic stability
- Tested for FDA/ISO 10993 and USP Class VI Biological Evaluation testing after Gamma and ETO sterilization
Eastman Tritan™ MX731
A high flow medical grade of Eastman Tritan™ that has viscosity reductions of 40-50% relative to Eastman Tritan™ Copolyester MX711
- Includes a vegetable-based mold release for clean processing
- Improved processability over traditional copolyesters
- Tested for FDA/ISO 10993 and USP Class VI Biological Evaluation testing after Gamma and ETO sterilization
Explore Tritan™ Applications for Your Business
Tritan™ is widely used in products such as reusable sports bottles, premium tableware, and small kitchen appliances.
We’re Here to Help
Complete the following fields with your information, as well as a short, detailed description of your request and a NAGASE Specialist will be in touch with you shortly.





















